Testimony | In Person
Event Title
Safeguarding Pharmaceutical Supply Chains in a Global Economy
October 30, 2019
- Testimony of
-
- Before the
Introduction
Madam Chairwoman, Ranking Member Burgess, and Members of the Subcommittee, I am Dr. Janet Woodcock, Director of the Center for Drug Evaluation and Research (CDER) at the Food and Drug Administration (FDA or the Agency), which is part of the Department of Health and Human Services (HHS).
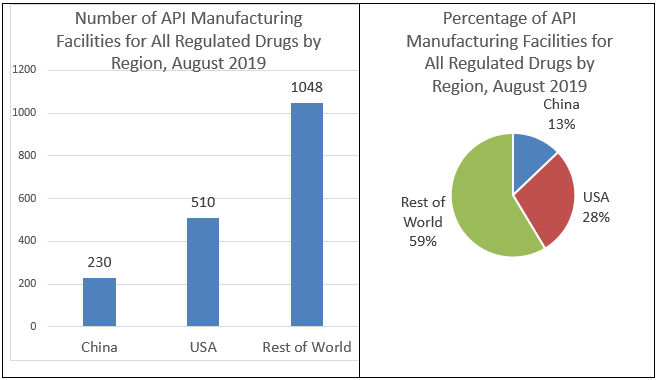
The United States, through its investment in biomedical research, has become a world leader in drug discovery and development, but is no longer in the forefront of drug manufacturing. Historically, the production of medicines for the U.S. population has been domestically based. However, in recent decades, drug manufacturing has gradually moved out of the United States. This is particularly true for manufacturers of active pharmaceutical ingredients (APIs), the actual drugs that are then formulated into tablets, capsules, injections, etc. As of August 2019, only 28 percent of the manufacturing facilities making APIs to supply the U.S. market were in our country. By contrast, the remaining 72 percent of the API manufacturers supplying the U.S. market were overseas, and 13 percent are in China. (See Figure 1) FDA’s data show that the number of registered facilities making APIs in China more than doubled between 2010 and 2019.
Figure 1: Manufacturing Sites of APIs for U.S. Market by Country or Region,
August 2019
While there are many reasons for this shift, underlying factors that are often cited include the fact that most traditional drug production processes require a large factory site, often have environmental liabilities, and can utilize a low-cost labor force. A 2009 paper by the World Bank, “Exploratory Study on Active Pharmaceutical Ingredient Manufacturing for Essential Medicines,” stated that if a typical Western API company has an average wage index of 100, this index is as low as 8 for a Chinese company and 10 for an Indian company.[1] China has lower electricity, coal, and water costs. Chinese firms are also embedded in a network of raw materials and intermediary suppliers, and so have lower shipping and transaction costs for raw materials. They also face fewer environmental regulations regarding buying, handling, and disposing of toxic chemicals, leading to lower direct costs for these firms. FDA’s 2011 report, “Pathway to Global Product Safety and Quality,” noted that both China and India enjoy a labor cost advantage and that API manufacturing in India can reduce costs for U.S. and European companies by an estimated 30 percent to 40 percent.[2]
Using traditional pharmaceutical manufacturing technology, a U.S.-based company could never offset the labor and other cost advantages that China enjoys simply by achieving higher productivity. However, FDA believes that advanced manufacturing technologies could enable U.S.-based pharmaceutical manufacturing to regain its competitiveness with China and other foreign countries, and potentially ensure a stable supply of drugs critical to the health of U.S. patients. Advanced manufacturing is a collective term for new medical product manufacturing technologies that can improve drug quality, address shortages of medicines, and speed time-to-market. Every field has a different set of production techniques that are considered advanced. Examples of some cross-cutting advanced manufacturing technologies include continuous manufacturing and 3D printing. Advanced manufacturing technology, which FDA supports through its Emerging Technology Program (ETP), has a smaller facility footprint, lower environmental impact, and more efficient use of human resources than traditional technology, as will be explained later in this testimony.
The pharmaceutical sector relies heavily on foreign sourcing for critical components, materials, and finished products, as identified in the U.S. Department of Commerce’s Office of Technology Evaluation’s 2011 report, “Reliance on Foreign Sourcing in the Healthcare and Public Health (HPH) Sector: Pharmaceuticals, Medical Devices and Surgical Equipment.”[3] However, use of foreign-sourced materials creates vulnerabilities in the U.S. drug supply. For example, in August 2018 FDA issued an alert that a Chinese API manufacturer, Sichuan Friendly Pharmaceutical Co. Limited, was recalling certain lots of porcine thyroid API due to inconsistent quality of the API.[4] This thyroid API comes from porcine (pig) thyroid glands and is used to make a medicine to treat hypothyroidism (underactive thyroid). FDA laboratory testing confirmed that the Sichuan Friendly API had inconsistent levels of active ingredients and should not be used to manufacture or compound drugs for patient use. Risks associated with over- or undertreatment of hypothyroidism could result in permanent or life-threatening adverse health consequences.
In December 2015, FDA alerted drug compounders that certain lots of baclofen API manufactured by Chinese manufacturer Taizhou Xinyou Pharmaceutical & Chemical Co., Limited might be at risk for contamination with particulates and should not be used to compound sterile injectable drugs. Taizhou manufactures APIs for repackagers and distributors, some of which sell these products to compounding facilities in the United States.[5]
FDA contacted Taizhou and the company confirmed that, due to the level of controls in the manufacturing process, the baclofen API it manufactures was not suitable for use in injectable drugs. FDA recommended that no baclofen API from Taizhou be used to manufacture or compound any injectable drugs. The affected API potentially could have posed serious safety risks for U.S. patients who used or received injectable drug products compounded with the affected baclofen, especially when administered directly into the spine (intrathecally). There was also a potential risk that the baclofen API might have been contaminated by endotoxin or microorganisms.
Today, I would like to share CDER’s information about the location of API manufacturing facilities in China, the United States, and the rest of the world; discuss the implications for national security; and explain how advanced manufacturing can increase the security and reliability of the U.S. drug supply.
Explanation of CDER’s Data and its Limitations
From a national security perspective, it is useful to look at the locations of facilities for three sets of drugs:
- All drugs on the U.S. market, including brand and generic drugs under approved applications, over-the-counter (OTC) drugs, and compounded medications.
- Drugs on the World Health Organization (WHO) Essential Medicines List that are marketed in the United States.
- Drugs on the medical countermeasures (MCM) lists. These include drugs we would use to counter biological, chemical, nuclear, or radiation threats and influenza.
CDER maintains a Site Catalog (“Catalog”) of all manufacturing facilities making drugs for the U.S. market, either through an approved application or that have registered and listed to supply drugs for the U.S market. This includes suppliers for API, finished dosage forms (FDF), or both. The APIs manufactured in these facilities may be used in prescription drugs (brand or generic), OTC drugs, and compounded drugs.
Data available to CDER have several limitations, including the following:
- Facilities listed in the Catalog may or may not be producing APIs. Including a facility in an application or the registration and listing process does not require a facility to produce API. Producing an API at the facility, or not producing it, is a business decision made by the company.
- Manufacturers are not required to report to FDA whether they are actually producing an API at a facility, and if they are, the volume they are producing.
- APIs made in listed facilities may be used in drugs for both the U.S. and other markets, and some APIs distributed in the United States are subsequently formulated into FDF that are then exported.
- Some FDF applications list more than one API supplier in the application. FDA has no visibility into which API supplier an FDF manufacturer uses at any given time.
- CDER has limited information about API suppliers for products that do not need an approved application from FDA to be marketed, such as compounded and OTC monograph drugs. API suppliers for such products may not register their facility with FDA if they are sending material to a drug product manufacturer outside the United States to make the FDF, which is then sold in the United States.
- Information in the Catalog is continually being updated. The analysis presented below is based on August 2019 listings and represents a snapshot at a point in time.
These limitations mean that, although CDER can describe the locations of API manufacturing facilities, we cannot determine with any precision the volume of API that China is actually producing, or the volume of APIs manufactured in China that is entering the U.S. market, either directly or indirectly by incorporation into finished dosages manufactured in China or other parts of the world.
API Manufacturing Facilities for All Regulated Drug Products
CDER’s analysis shows that overall, China has only a modest percentage of the facilities able to produce APIs for the U.S. market. For all regulated drugs, China has 230 (13 percent) of the API manufacturing facilities, while the United States has 510 (28 percent), and the rest of the world has 1048 (59 percent). “All regulated drugs” includes prescription (brand and generic), OTC, and compounded drugs. (See Figure 2) However, the percentages of APIs produced at these facilities may differ, and as mentioned above, cannot be determined from the data available to FDA.
Figure 2: Number and Percentage of API Manufacturing Facilities for All Drugs by Region, August 2019
API Manufacturing Facilities for WHO Essential Medicines on the U.S. Market
The 2019 WHO Essential Medicines List comprises 461 drugs that have been selected by the WHO Expert Committee to meet the most important needs in a health system. This list includes application and non-application products across a wide range of therapeutic categories such as anesthetic, antibacterial, antidepressant, antiviral, cardiovascular, anti-diabetic, and gastrointestinal agents.
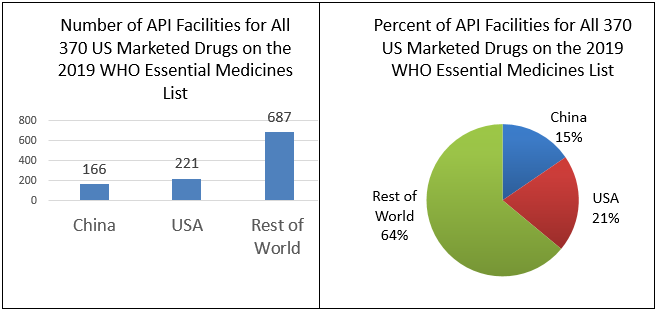
FDA matched 370 of the drugs on the WHO Essential Medicines List with products listed for the U.S. market and determined the location of the facilities used to make their APIs.[6] FDA data show that there is a total of 1,079 API facilities worldwide that make the 370 drugs on the WHO list that are marketed in the U.S. Of these, 166 (15%) are in China, 221 (21%) are in the United States, and 687 (64%) are in the rest of the world. (See Figure 3)
Figure 3: Number and Percentage of API Facilities for the 370 U.S. Marketed Drugs on the 2019 WHO Essential Medicines List
FDA determined that there are three WHO Essential Medicines whose API manufacturers are based only in China. The three medicines are: capreomycin and streptomycin, both indicated to treat Mycobacterium tuberculosis; and sulfadiazine, used to treat chancroid and trachoma.
The distribution of API facilities worldwide varies from drug to drug and may differ from the patterns for all drugs or WHO Essential Medicines List Drugs.
API Manufacturing Facilities for Medical Countermeasures
FDA maintains a list of drugs that are used as medical countermeasures (MCMs) against threats in four categories: biological threats, chemical threats, influenza, and radiation threats. Many of these drugs are contained in strategic drug stockpiles, including the Nation’s Strategic National Stockpile, the Nation’s largest supply of potentially life-saving pharmaceuticals and medical supplies for use in a public health emergency severe enough to cause local supplies to run out.
For APIs for 14 drugs in the biological threat category, China has 37 facilities, the United States has 19, and the rest of the world has 117. China has just six of the facilities producing APIs for the 10 drugs in the chemical threat category, versus 24 in the United States and 52 in the rest of the world. (See Table 1)
| MCM Type (# products) |
U.S. API Sites # (%) |
China API Sites # (%) |
Other Foreign API Sites # (%) |
|---|---|---|---|
| Biological (14) | 19 (11%) | 37 921% | 117 (68%) |
| Chemical (10) | 24 (29%) | 6 (7%) | 52 (64%) |
| Influenza (3) | 2 (11%) | 0 (0%) | 16 (89%) |
| Radiation (7) | 13 (46%) | 0 (0%) | 15 (54%) |
Table 1: Number and Percent of API Manufacturing Sites for MCM Drugs for Use Against Biological, Chemical, and Radiation Threats, and Influenza, August 2019
China has none of the facilities making APIs for medicines to prevent or treat influenza versus two in the United States and 16 in the rest of the world. China also has none of the facilities producing APIs for radiation threats. The United States has 13 of these facilities versus 15 in the rest of the world.
Ciprofloxacin and doxycycline are two drugs considered critical as MCMs and used to treat anthrax and plague. As shown in Table 2 below, the United States has one facility for ciprofloxacin, versus three in China and 21 in other foreign countries. The United States has fewer facilities than China or other foreign countries for doxycycline. (See Table 2)
| MCM | # (%) U.S. API Sites | # (%) China API Sites | # (%) Other Foreign API Sites |
|---|---|---|---|
| Ciprofloxacin | 1 (4%) | 3 (12%) | 21 (84%) |
| Doxycycline | 2 (18%) | 3 (27%) | 6 (55%) |
Table 2: Number and Percent of API Manufacturing Sites for Ciprofloxacin and Doxycycline by Region, August 2019
Implications for National Security
The security of the nation’s drug supply rests on three main factors: freedom from dependence on foreign sources of API, the resilience of our domestic manufacturing base, and the reliability of the facilities that make products for the U.S. market.
Dependence: How dependent are we on China, India, or other countries for the APIs used in drugs produced for patients in the United States? How has this dependence changed over time?
The number of Chinese facilities producing APIs for the U.S. market has increased over the past decade, as part of a massive movement of pharmaceutical production offshore. This movement is being driven by the pharmaceutical industry’s desire for cost savings and less stringent environmental regulations. Absent any intervention, FDA believes that this trend is likely to continue.
However, data available to FDA do not enable us to calculate the volume of APIs being used for U.S.-marketed drugs from China or India, and what percentage of U.S. drug consumption this represents. As mentioned above, we do not know whether Chinese facilities are actually producing APIs, how much they are producing, or where the APIs they are producing are being distributed worldwide, including in the United States.
Resilience: How resilient is the U.S. manufacturing base? How quickly could U.S.-based manufacturers increase their production of APIs to meet domestic demand if China or India, or another country, ceased supplying the United States, particularly for drugs on the WHO Essential Medicines list or a subset that is widely used by the U.S. population?
To answer this question, FDA would need to know:
- how much unused capacity exists in the U.S. manufacturing base for APIs;
- how much additional API this capacity could supply within a given time period;
- how far this capacity would go in filling the gap between U.S. patients’ needs and the amount available if China or India, or another country, were to reduce or stop the supply to the U.S. market; and
- how long would it take to increase production enough to meet patients’ needs, and whether the financial investment would be sustainable for the pharmaceutical industry.
Since we do not currently know whether API manufacturing facilities are actually producing the drug, or in what volume, or what portion of U.S. drug consumption is dependent on APIs from China or India, or another country, we cannot perform a reliable gap analysis.
Even if we could estimate the potential API shortfall and available production capacity, pharmaceutical companies make business decisions about whether to produce a drug product, including an API, and FDA does not have the power to tell them to make a drug. This provides additional uncertainty in assessing the potential responsiveness of the U.S. manufacturing base to a crisis triggered by another country’s withdrawal.
Reliability: How reliable is the manufacturing base that produces APIs for the U.S. market?
FDA recently analyzed 163 drugs that went into shortage during the five-year period from 2013 to 2017 and found that quality problems were responsible for the shortages 62 percent of the time.[7] These shortages can worsen patients’ health outcomes by causing delays in treatment or changes in treatment regimens, such as substituting less-effective or well-tolerated medicines when a drug of choice is not available.
In looking for ways to ensure Americans’ access to a supply of safe and effective drugs, we need to consider all three dimensions of the problem.
Summary of National Security Findings
FDA’s information shows that overall, the number of China’s API facilities is somewhat smaller than the United States, but comparable in size and growing. However, because of the limitations of available data, we cannot assess the extent of U.S. dependence on China. For instance, we do not have information about the volume of API being produced in China or even in the United States, or how much of China’s API output reaches the U.S. market through other countries.
Similarly, we do not have information that would enable us to assess the resilience of the U.S. manufacturing base, should it be tested by China’s withdrawal from supplying the U.S. market. We do know that the U.S. drug supply is being compromised by drug shortages, in most cases triggered by manufacturing quality problems by U.S.-based as well as foreign producers.
Advanced Manufacturing Offers a Multi-dimensional Solution
Advanced manufacturing is the use of innovative technology to improve products and processes. Although widely used in some other industries, such as automotive, aerospace, and semiconductors, advanced manufacturing is now just beginning to be used by pharmaceutical companies. For API and/or FDF manufacturing, new technologies include “continuous manufacturing” (CM), wherein the finished drug product is produced as a continuous stream, as opposed to traditional batch manufacturing where breaks or stops exist between different processing steps. In some examples of advanced pharmaceutical manufacturing, production can be continuous from chemical synthesis of the active ingredient through production of the tablets or other dosage forms.
Advanced manufacturing offers many advantages over traditional pharmaceutical manufacturing, and if the United States invests in this technology, it can be used to reduce the Nation’s dependence on foreign sources of APIs, increase the resilience of our domestic manufacturing base, and reduce quality issues that trigger drug shortages or recalls. For example:
- Product quality can be precisely controlled with modern automation and control systems and can be closely monitored during production by using high-resolution analytics.
- High technology, computer-controlled production facilities are better able to rapidly respond to changes in demand because they typically do not have the equipment scale-up issues associated with traditional methods and can be capable of seamlessly producing a variety of dosages and even dosage forms.
- Advanced manufacturing platforms also have a much smaller footprint than traditional manufacturing platforms, and the equipment can be made portable so that it can be moved closer to markets, reducing the need for transcontinental shipping of components.
- Medicines can be produced at lower cost than by traditional methods.
- Environmental impact of manufacturing is significantly reduced.
By supporting the growth of advanced manufacturing in the United States, we can reduce our dependence on China and other overseas manufacturers for APIs as well as improve the resilience and responsiveness of our manufacturing base and reduce drug shortages.
FDA’s advanced manufacturing initiative is fostering this growth in several ways.
Emerging Technology Program (ETP)
The ETP, launched in late 2014, encourages and supports the adoption of innovative technology to modernize pharmaceutical development and manufacturing through close collaboration with industry and other relevant stakeholders starting from early technology development.
To reduce barriers to entry for advanced manufacturing, the Emerging Technology Team (ETT) provides a gateway for the early (pre-submission) discussion of innovative technologies and approaches, even before a candidate drug is identified. The ETT supports the entry, assessment, and lifecycle management of advanced manufacturing at CDER. It provides subject matter experts and fosters coordination within CDER and FDA’s Office of Regulatory Affairs (ORA) for precedent-setting issues regarding quality and good manufacturing practices. ETT serves as a hub for identification of application-driven regulatory and research needs and provides strategic input for supporting advanced manufacturing innovation. Based on ETT efforts in continuous manufacturing, CDER’s Office of Pharmaceutical Quality (OPQ) published a draft guidance, “Quality Considerations for Continuous Manufacturing” of solid oral dosage forms in early 2019.[8]
Under this program, CDER has approved five drug applications utilizing continuous manufacturing for FDF manufacturing, and the first application utilizing 3-D printing technologies. Currently, these drugs are being made in the United States, and one drug is being made both in the United States and in the United Kingdom.
Regulatory and Policy Initiatives
The adoption of advanced manufacturing technologies may pose a challenge to the current regulatory framework, because most regulations were developed based on traditional batch manufacturing methods under a unified pharmaceutical quality system. As a result, FDA has launched an effort to identify and implement needed changes in the regulatory structure. For example, new policy and regulatory topics related to emerging technologies include the management of data-rich environments, the evolving concepts of process validation for advanced manufacturing systems, and the regulatory oversight of post-approval changes for such systems. Furthermore, CDER, in collaboration with the Biomedical Advanced Research and Development Authority (BARDA), is working on a strategy and new regulatory framework to develop and implement miniature, mobile manufacturing platforms (“Pharmacy on Demand”) for manufacture of essential drugs near or at the point of care.
FDA actively engages with stakeholders in industry, academia, and other regulatory agencies to identify and address regulatory hurdles to adoption of advanced manufacturing. For example, CDER, in partnership with FDA’s Center for Biologics Evaluation and Research (CBER), is leading the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) effort to develop the Q13 guideline on continuous manufacturing of drug substances and drug products for both small-molecule and biological products, which will help to achieve global regulatory harmonization.
Intramural and Extramural Research
Laboratories in CDER’s OPQ actively conduct advanced manufacturing research and invest in equipment, facilities, and personnel with expertise to investigate these topics. OPQ has established the Center of Excellence for Manufacturing Science and Innovation to coordinate internal advances in manufacturing research for both small molecules and biologics. OPQ publishes and leads research on continuous manufacturing, advanced analytics for process controls, and modeling and simulation. OPQ also provides training for assessment and inspection personnel.
Extramurally, OPQ awards research grants and contracts for advanced manufacturing and emerging technologies. OPQ also participates in consortia with academia and industry to identify new areas for research in advanced manufacturing. Participation in these intramural and extramural research efforts occurs with strong alignment and coordination between assessment, policy, and surveillance offices. This ensures that resources are allocated to projects that provide practical approaches to regulating innovative technologies.
On behalf of FDA, I would like to thank Congress for having the foresight to provide resources to support our efforts to assist the pharmaceutical industry as it makes a transition from traditional manufacturing methods to the use of advanced technologies. These new tools and methods have the potential to reinvigorate our pharmaceutical manufacturing base and repatriate it from overseas. This will help to ensure that Americans have a secure and reliable supply of medicines in the future, as well as contribute to our economy.
Promoting Domestic Manufacturing
In FY19, Congress approved appropriations to promote domestic manufacturing with the intent that FDA advance modern drug and biological product technologies. The spending plan for these additional resources builds on the CDER strategic goals to improve overall staff understanding and expertise in advanced manufacturing by expanding support of these innovative technologies in assessment, policy, surveillance, and research, as well as by making programmatic improvements. In addition, this funding is used to reinforce extramural outreach with stakeholders via planned technology forecasting activities with the National Academies of Sciences, Engineering, and Medicine and other forums.
Conclusion
The increasing number of API manufacturing sites in China and other countries suggests that the United States’ reliance on Chinese and other foreign sources of API is growing. FDA has been working diligently in collaboration with industry and other federal agencies to ensure our reliance of foreign manufacturing does not pose a national security risk. While FDA cannot tell industry where they can and cannot manufacture APIs, we can work with industry to utilize new technologies and new manufacturing methods to further incentivize domestic production of drugs and APIs. These new ways of making drugs could, with the proper strategies, revitalize pharmaceutical manufacturing in the United States.
Footnotes
1. Bumpas, Janet; Betsch, Ekkehard. Exploratory study on active pharmaceutical ingredient manufacturing for essential medicines (English). Health, Nutrition and Population (HNP) discussion paper. Washington, DC: World Bank. Exploratory study on active pharmaceutical ingredient manufacturing for essential medicines. Accessed September 30, 2019.
2. U.S. Food and Drug Administration, “Pathway to Global Product Safety and Quality,” A Special Report, p. 20. Accessed October 4, 2019.
3. See Reliance on Foreign Sourcing in the Healthcare and Public Health (HPH) Sector: Pharmaceuticals, Medical Devices, and Surgical Equipment, accessed October 15, 2019.
4. See FDA alerts drug makers of a recall of porcine thyroid API from Sichuan Friendly Pharmaceutical Co., Limited, China, accessed October 6, 2019.
5. See FDA Warns of potential contamination of baclofen active pharmaceutical ingredient from Taizhou Xinyou Pharmaceutical & Chemical Co., Limited, China, accessed October 7, 2019.
6. Some of the 461 drugs on the 2019 WHO Essential Medicines List were excluded from the analysis because they are not regulated by CDER. The List includes products regulated by the Center for Biologics Evaluation and Research (CBER), such as cholera vaccine and anti-rabies immunoglobulin, and products regulated by the Center for Devices and Radiological Health (CDRH), such as diaphragms and condoms.
7. U.S. Food and Drug Administration, “Drug Shortages: Root Causes and Potential Solutions,” October 2019.
8. Quality Considerations for Continuous Manufacturing, Draft Guidance for Industry, accessed October 15, 2019.