Testimony | In Person
Event Title
COVID-19 and Beyond: Oversight of the FDA's Foreign Drug Manufacturing Inspection Process
June 2, 2020
- Testimony of
-
Mark Abdoo
- Before the
Chairman Grassley, Ranking Member Wyden, and Members of the Committee, thank you for the opportunity to testify today on a matter of the utmost importance to the Agency: protecting the safety, quality, and availability of medicines for Americans.
The U.S. drug supply is among the safest in the world. FDA thoroughly reviews drug applications to ensure that medications are safe and effective before they reach the market and oversees drug quality post-approval. The Agency inspects drug manufacturing facilities located around the world with comparable depth and rigor based on an assessment of risk to public health. FDA laboratories test for drug quality, using testing standards set by the United States Pharmacopeia, or standards submitted in marketing applications, or methods developed by FDA. This testing has consistently shown that medicines manufactured in foreign countries that are imported into the United States meet U.S. market quality standards. When FDA identifies significant manufacturing or safety issues, it quickly acts to protect Americans.
During the COVID-19 pandemic, FDA is continuing to utilize and implement additional alternative inspection tools and approaches while postponing foreign and domestic routine surveillance facility inspections. This will continue as conditions warrant, with the exception of certain mission critical inspections, including pre-approval and for-cause assignments. Mission critical inspections are identified on a case-by-case basis and conducted with appropriate safety measures in place.
Importantly, during this interim period we’re evaluating additional ways to conduct our inspectional work that would not jeopardize public safety and protect both the firms and the FDA staff. This can include, among other things, evaluating records in advance of or in lieu of conducting an onsite inspection when travel is not permissible, when appropriate. We want to assure the American public that we have full confidence in the safety and quality of the products we all use every day and that the FDA will continue to leverage all available authorities to continue to ensure the integrity of the products we regulate.
Today we will provide the Committee with an overview of the history of FDA’s foreign drug inspection program, and the ways it has evolved in response to the industry’s globalization and changes in law and regulation. We will also explain our approach when our inspections indicate that a facility does not operate in keeping with established quality standards. These standards are known as current good manufacturing practices (CGMPs). We will also describe some potential enhancements that would enable FDA to complement our foreign drug inspection program. The Agency believes that over the longer term, we should encourage investment in advanced manufacturing technology and in strengthening the approach by which manufacturers assure the quality of their products. This approach, which we call quality management maturity, would provide a safer and more secure drug supply because it can help prevent many quality problems from occurring in the first place. Advanced technology, which can be more cost-effective and environmentally friendly than traditional manufacturing technology, may also enable the United States to play a larger role in pharmaceutical manufacturing.
The Globalization of Pharmaceutical Manufacturing
Over the past 30 years, pharmaceutical manufacturing has become an increasingly global enterprise. Beginning in the 1970s, industry moved away from the mainland U.S., first to Puerto Rico in response to tax incentives, and then to Europe and nations that were developing at the time, such as China and India. Developing nations can provide significant cost savings to pharmaceutical companies because of their lower labor, energy, and transportation costs. In addition, they often have weaker environmental regulations than more developed countries. A World Bank study estimated that in 2004, China and India held a cost advantage of about 40 percent when compared with the U.S. and Europe. 1 FDA’s 2011 report, “Pathway to Global Product Safety and Quality,” also noted that both China and India enjoy a labor cost advantage and that manufacturing active pharmaceutical ingredients (APIs) in India can reduce costs for U.S. and European companies by an estimated 30 percent to 40 percent. 2
As the U.S. drug market shifted toward lower-priced generic drugs, manufacturers came under increasing cost pressure and found these efficiencies compelling reasons to locate more of their facilities overseas, particularly in developing parts of the world. This shift is reflected in the Center for Drug Evaluation and Research’s (CDER’s) Site Catalog (“Catalog”), which lists all drug manufacturing facilities worldwide that are subject to routine FDA inspections. 3 As of May 2020, 26 percent of facilities manufacturing APIs and 46 percent of the facilities producing finished dosage forms (FDFs) of human drugs for the U.S. market were located in the U.S. (See Figures 1 and 2)
This movement accelerated in the 2000s, but due to statutory mandates for biennial domestic inspections and limited staffing, FDA’s inspectorate remained focused on domestic manufacturing. Until passage of the Food and Drug Administration Safety and Innovation Act (FDASIA) in 2012 (P.L.112-144), the Agency was legally required to inspect manufacturing facilities in the U.S. every two years but had no similar mandate for the inspection frequency of foreign facilities. This resulted in more frequent inspections for domestic facilities.
The Globalization of FDA’s Drug Inspection Program
In response to the move from domestic to global manufacturing and the passage of FDASIA, FDA developed and implemented a comprehensive strategy to facilitate greater coordination and oversight of medical products. In addition to increasing foreign inspections, our efforts have included:
- developing new enforcement and regulatory tools
- increasing collaboration with foreign regulators and other stakeholders
- developing internationally-harmonized standards and standards convergence
- educating foreign industry about FDA requirements, and
- increasing transparency and accountability in the supply chain
- establishing foreign offices with an overseas footprint in China, India, Europe, and Latin America.
Responsibility for addressing these global issues is distributed across the agency. The Office of Regulatory Affairs (ORA) conducts inspections and reviews imported products offered for entry into the United States. FDA’s product centers focus on international policy and outreach that touches on their portfolio of regulated products. The Office of Global Policy and Strategy serves as a focal point for FDA-wide coordination and information-sharing and a point of access to multilateral organizations; addresses issues related to international trade of regulated products and mutual recognition agreements; enters into arrangements that facilitate the sharing of information with global regulatory counterparts; and manages FDA’s foreign offices around the world.
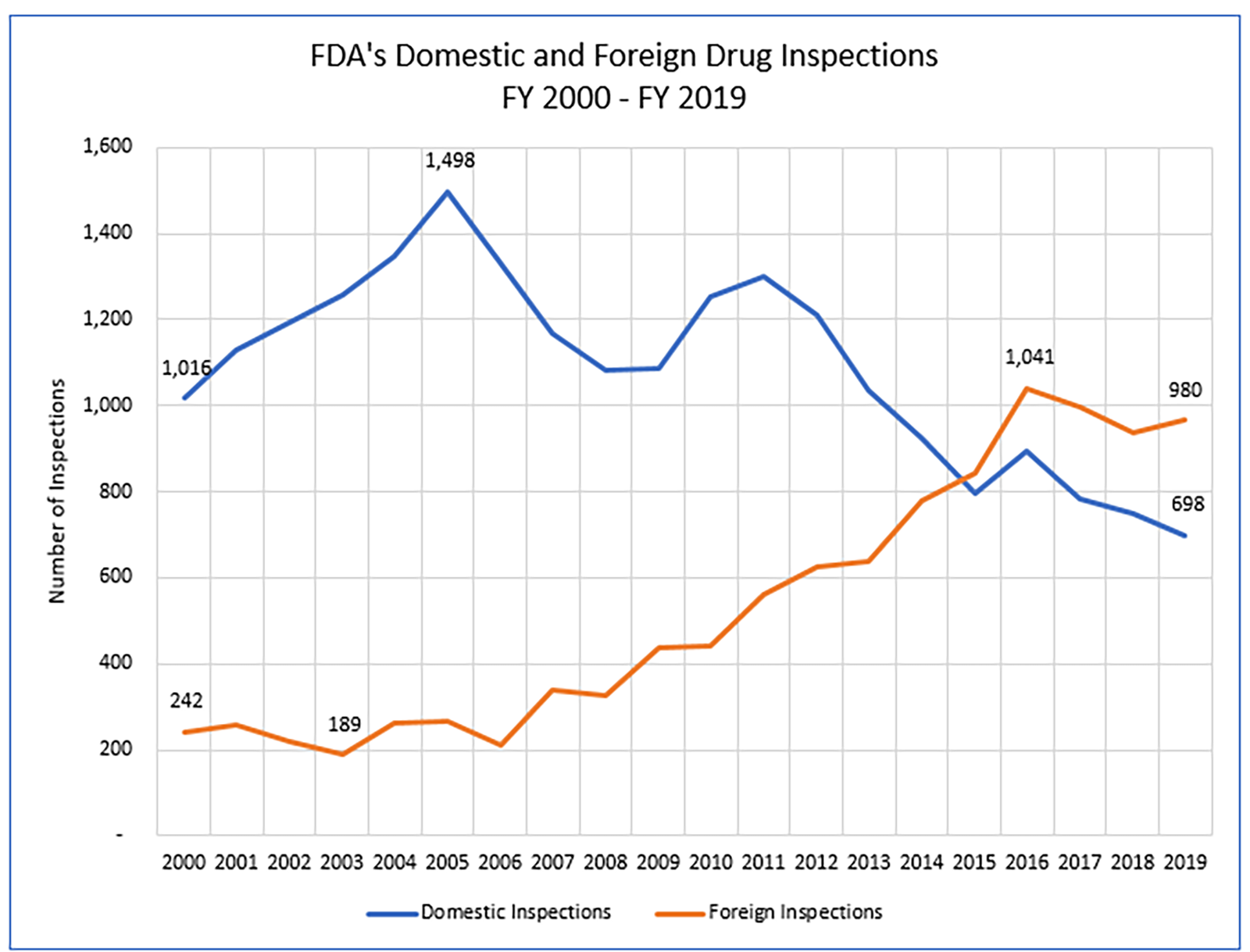
FDA’s drug inspection program shifted from one focused heavily on U.S.-based facilities through the early 2000s to a program that, since 2015, has conducted more foreign than domestic drug inspections. (See Figure 3.) FDA’s drug inspection program is now risk-based. FDA prioritizes for inspection facilities deemed higher-risk based on specific, defined criteria.
Figure 3: FDA’s Inspections of Foreign Drug Manufacturing Facilities Increased Sharply After 2006 and Have Exceeded Inspections of Domestic Drug Facilities Since 2015
Types of Inspections
The types of inspections performed in both domestic and foreign facilities include pre-approval, surveillance, and for-cause inspections.
- Pre-approval inspections: conducted as part of the review of an application to market a new brand or generic drug.
- Surveillance inspections: Used to monitor the manufacturing process and the quality of distributed drugs. FDA uses the findings to evaluate whether a manufacturer is complying with CGMPs. In general, the Agency does not announce domestic surveillance inspections to the company in advance but usually announces foreign surveillance inspections in advance, partly due to logistics such as arranging travel and access to facilities, securing visas, and partly because of the high costs of conducting foreign inspections. Whether inspections are announced often depends on particular cases and the history of specific facilities.
- For-cause inspections: Triggered when FDA has reason to believe that a facility has serious manufacturing quality problems or when FDA wants to evaluate corrections that have been made to address previous violations. For-cause inspections can be announced or unannounced, whether domestic or international, depending on the specific situation.
When the Agency has determined the need to do an unannounced inspection, FDA has conducted such operations. Over the past several years, FDA investigators have conducted unannounced inspections at foreign manufacturing facilities, including in India and China. When significant issues are uncovered at a foreign manufacturing facility, the Agency uses additional tools to protect patients including placing the facility on import alert, which is used to prevent potentially violative products from entering the U.S. market.
The Site Selection Model
To address the need to prioritize use of limited resources, in 2005 FDA implemented a risk-based approach to drug facility surveillance inspections. A mathematical model, the Site Selection Model (SSM), was designed to select facilities with the greatest potential for public health risk should they not comply with established manufacturing quality standards. FDA uses results of the model to prepare a prioritized list of facilities for inspection.
The passage of FDASIA ratified our risk-based approach and removed the requirement to inspect domestic facilities on a fixed biennial schedule. FDASIA also enhanced our inspectional authority by requiring facilities to provide, upon request, records or other information in lieu of or in advance of an inspection. Additionally, under another provision added by FDASIA, if the owner or operator of a foreign facility delays, denies, or refuses to permit inspection, all drugs manufactured at that facility would be deemed “adulterated.” 4 The Agency thanks Congress for enacting this law.
In 2007, FDA began planning the shift of its investigator workforce to cover foreign facilities and to balance allocation between domestic and foreign inspections. Both the Generic Drug User Fee Amendments (GDUFA) of 2012 and its reauthorization in 2017 provided new resources to FDA for inspecting foreign facilities, which as we have noted are often the source for APIs and FDFs of generic drugs.
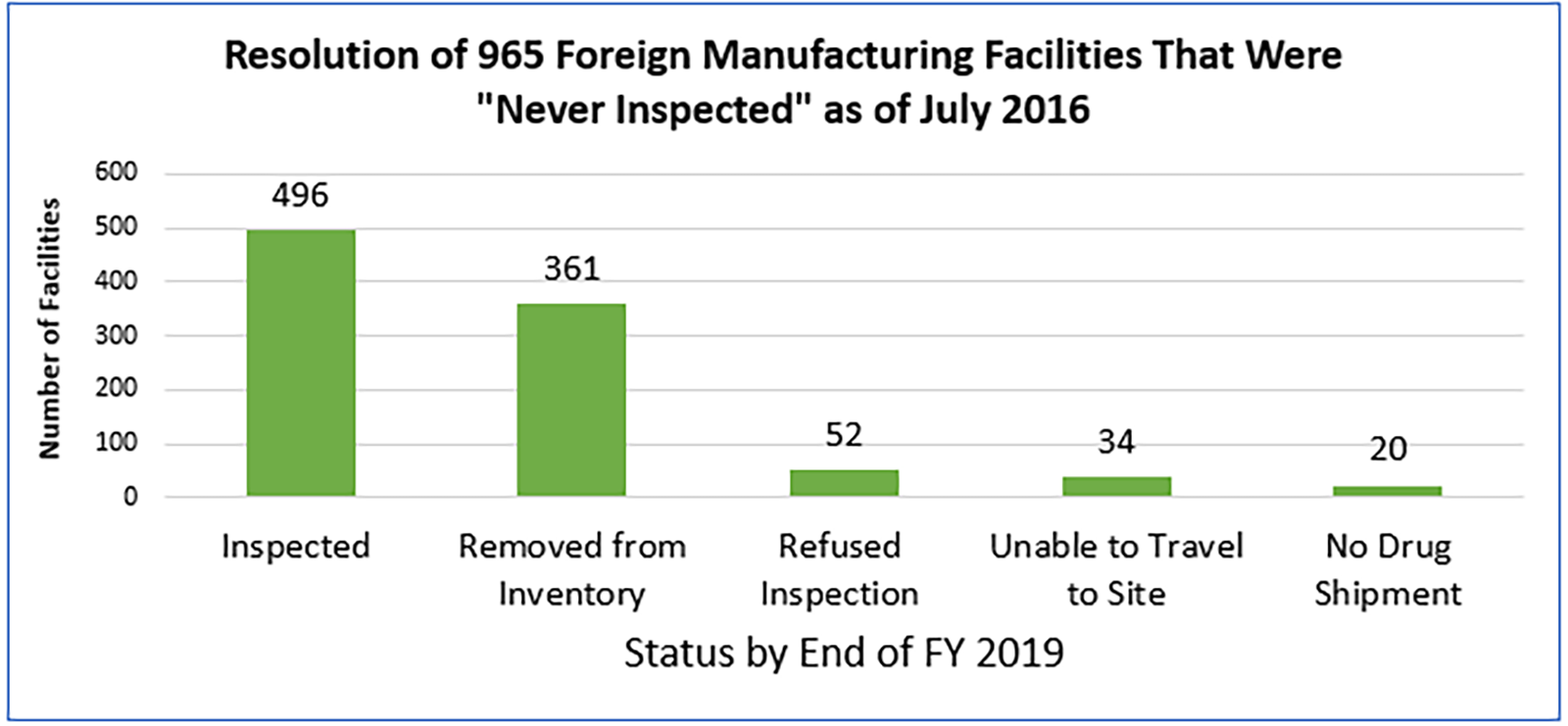
With new resources, FDA has been able to inspect some facilities that previously had not been inspected. Catalog showed that as of December 2016, there were 963 foreign manufacturing facilities that had never been inspected by FDA. All of the 963 5 foreign manufacturing facilities that GAO reported to be uninspected (as of December 2016) have now been addressed. By the end of FY 2019, FDA had inspected 496, or approximately 52 percent, of these previously uninspected facilities (see Figure 4). An additional 361 facilities (37 percent) were removed from the Catalog because they were no longer part of FDA’s inspection obligations for a number of reasons, e.g., they had gone out of business, were not serving the U.S. market, or had been registered with FDA erroneously. In addition, 52, or five percent, of the facilities had refused inspection; 6 34, or four percent, of the facilities were inaccessible to FDA investigators because they were unable to travel to them (e.g., as a result of travel warnings); and 20, or two percent, had no imported drug shipments to the U.S.
Figure 4. FDA has now evaluated all previously “never inspected” facilities
The SSM is at the core of FDA’s surveillance inspection prioritization program and ensures a uniform approach for domestic and foreign facility inspections. The Agency uses the model to calculate a score for every facility in its Catalog using risk-based factors. Factors in the SSM include:
- Inherent product risk. Different types of products carry different levels of risk based on characteristics such as dosage form, route of administration, or whether the product is intended to be sterile. For example, a manufacturing facility that makes sterile injectable drug products will have a higher inherent product risk than a facility that makes oral capsules.
- Facility type. Risk levels can vary depending on the operations that a facility performs. A facility that manufactures drug product or active ingredients is higher in risk than a facility that only packages drug product.
- Patient exposure. The more products a facility manufactures, the more likely a patient is to encounter products made at that facility. This refers to both number and types of products manufactured. A facility that manufactures many products will have a higher exposure factor than a facility that makes few products.
- Inspection history. A facility that has not met established quality standards when previously inspected is considered higher risk than those that have met standards in the past.
- Time since last inspection. As the time since a facility was last inspected increases, the risk that it may not meet established quality standards increases, as does the need for re-inspection.
- Hazard signals. Events such as product recalls or manufacturers’ or patients’ reports of quality problems associated with a facility increase the risk score when compared with facilities that have fewer or no major hazard signals.
FDA compares a facility’s score to others in the Catalog and ranks them by risk, with the highest risk assigned for inspection regardless of location.
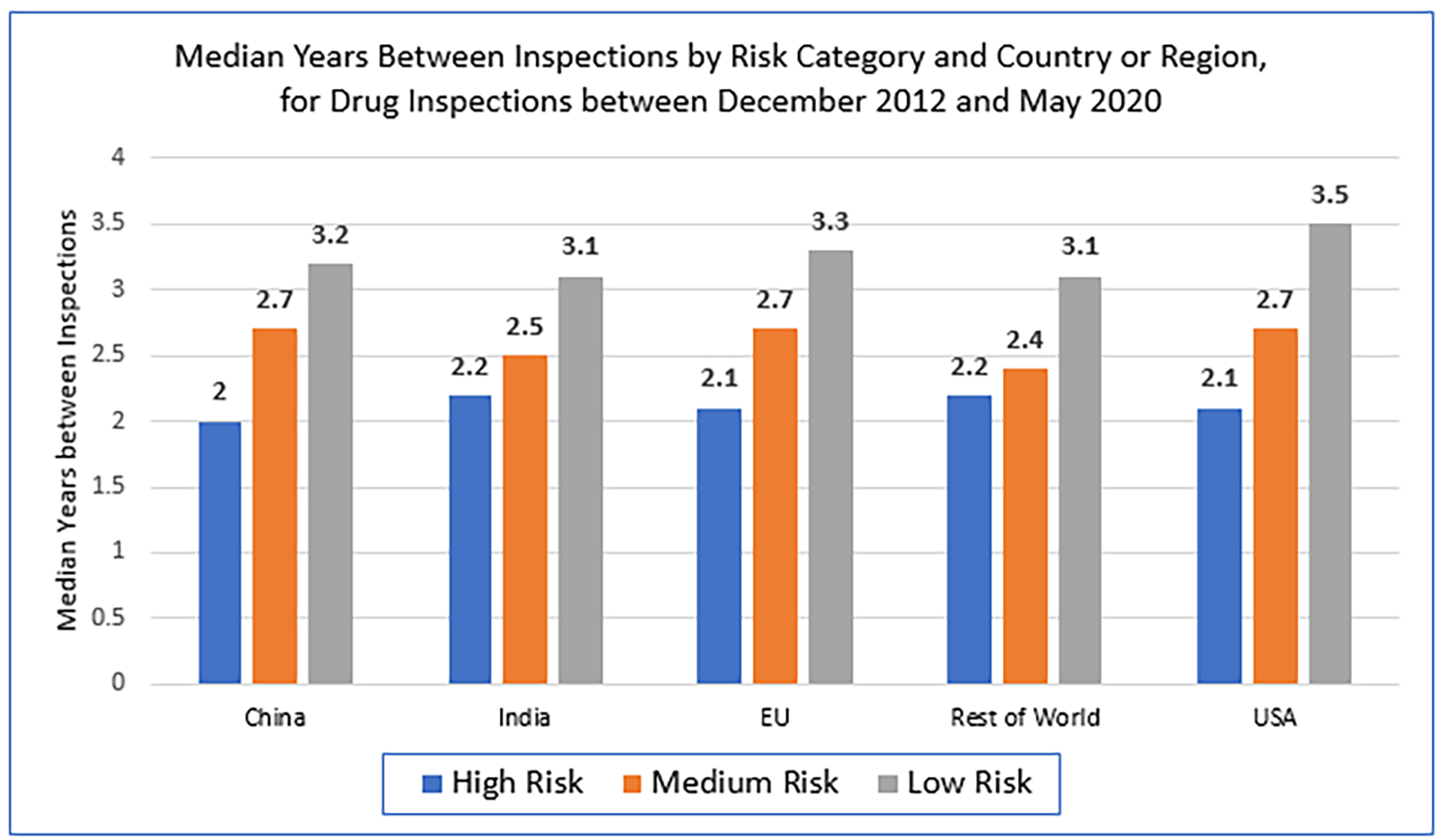
If the three factors that are fairly static for a facility (inherent product risk, facility type and patient exposure) are used to risk rank facilities, for inspections conducted from December 2011 to May 2020, the median time between inspections was 2.1 years for high-risk facilities. In general, all high-risk facilities were inspected with about the same frequency regardless of location. (See Figure 5)
Figure 5. FDA inspected high-risk manufacturing facilities more frequently than medium- or low-risk facilities, and medium-risk facilities more frequently than low-risk facilities, across all countries or regions. In general, all facilities in a risk category were inspected with about the same frequency, regardless of location.
Inspection Outcomes
Following inspection of a manufacturing facility, FDA classifies the inspection as “no action indicated” (NAI), “voluntary action indicated” (VAI), or “official action indicated” (OAI).
- No Action Indicated (NAI) means that no objectionable conditions or practices (e.g., quality problems) were found during the inspection (or they were minor problems that do not justify further regulatory action).
- Voluntary Action Indicated (VAI) means objectionable conditions or practices were found but the Agency is not prepared to take or recommend any administrative or regulatory action.
- Official Action Indicated (OAI) means regulatory and/or administrative actions will be recommended. 7
Not surprisingly, with more frequent inspections directed to higher-risk facilities since 2012, FDA uncovered more deficiencies, particularly in foreign facilities that had not been inspected as frequently as domestic ones prior to the inception of FDASIA and GDUFA. Notably these were foreign inspections that were generally announced to facilities in advance (pre-announced). Nevertheless, 90 percent or more of the final outcomes of inspections were acceptable (NAI or VAI) in all countries or regions except India (See Figure 6).
Figure 6. The majority of final inspection outcomes for manufacturing facilities making human drugs were acceptable, meaning that they were classified as having No Action Indicated or Voluntary Action Indicated. However, India had a lower percentage of acceptable outcomes than other countries and regions. (These were outcomes as of May 2020 for the most recent inspection of facilities within FDA’s Catalog.)
Both foreign and domestic drug manufacturers must meet the same regulatory requirements in terms of complying with established quality standards (CGMPs). If a facility doesn’t meet CGMP standards upon inspection, FDA has an array of regulatory tools it can use to encourage a company to remediate their manufacturing processes and achieve compliance. These tools include warning letters, import alerts, injunctions, and seizures. 8 If the Agency observes on a follow-up inspection that a facility still does not meet CGMP standards, it can escalate the matter as appropriate.
If a foreign facility is found to have quality problems serious enough for FDA to classify it as OAI, the Agency can place a facility on Import Alert, which is used to prevent potentially violative drugs from the facility from entering the U.S. Generally, FDA will remove a facility from a CGMP-related Import Alert after an onsite re-inspection demonstrates that the problems have been remediated and the firm is in compliance with CGMP.
Despite the tools at FDA’s disposal, we still face some challenges in ensuring the safety of imported drugs entering our drug supply. Current mandates for facility inspection prior to import or marketing of a drug in the U.S. are typically in the context of premarket approval requirements. For drugs that are subject to premarket approval requirements, FDA has an opportunity to evaluate and inspect the manufacturing facilities as part of the application review process. However, for drugs that are not subject to premarket approval requirements, manufacturers may not be subject to FDA inspection before such products are shipped to or distributed in the U.S. Drugs in this category typically include OTC monograph drugs and APIs used in pharmacy compounding. FDA may be required to engage in more challenging and resource intensive efforts to identify and respond to any problems that arise subsequently; however, patients may have already been exposed to the drugs. For example, in 2019 we issued a warning letter to a discount retailer for receiving OTC drugs produced by foreign manufacturers with serious violations of CGMPs. The majority of the foreign facilities involved had distributed drugs to the U.S. prior to FDA inspections. 9
FDA’s Program Alignment Initiative and Concept of Operations Agreement
The inspection of drug manufacturing facilities relies on the collaboration of two organizations within FDA: ORA, which includes the field force of investigators who conduct the inspections, and CDER, which includes policy and regulatory experts who establish the policies governing drug quality, assess risks, and review action recommendations, including OAI recommendations from ORA and for-cause inspections to determine the final classification and whether appropriate regulatory action is required. CDER also includes assessors who evaluate applications for marketing approval and post-marketing changes. In May 2017, as part of a broader agency initiative called Program Alignment, ORA implemented a program-based management structure aligning staff by FDA-regulated product. This created a specialized inspectorate focused on human drugs.
FDA modeled its oversight of the increasingly complex and global drug manufacturing supply chain to better integrate facility evaluations and inspections for human drugs – to improve our efficiency, reach, and the public health. In June 2017, CDER and ORA entered into a Concept of Operations 10 (ConOps) agreement to more effectively manage the growing complexity of the pharmaceutical landscape. The agreement, Integration of FDA Facility Evaluation and Inspection Program for Human Drugs: A Concept of Operations, outlines the responsibilities and the workflow for pre-approval, surveillance, and for-cause inspections at domestic and international facilities.
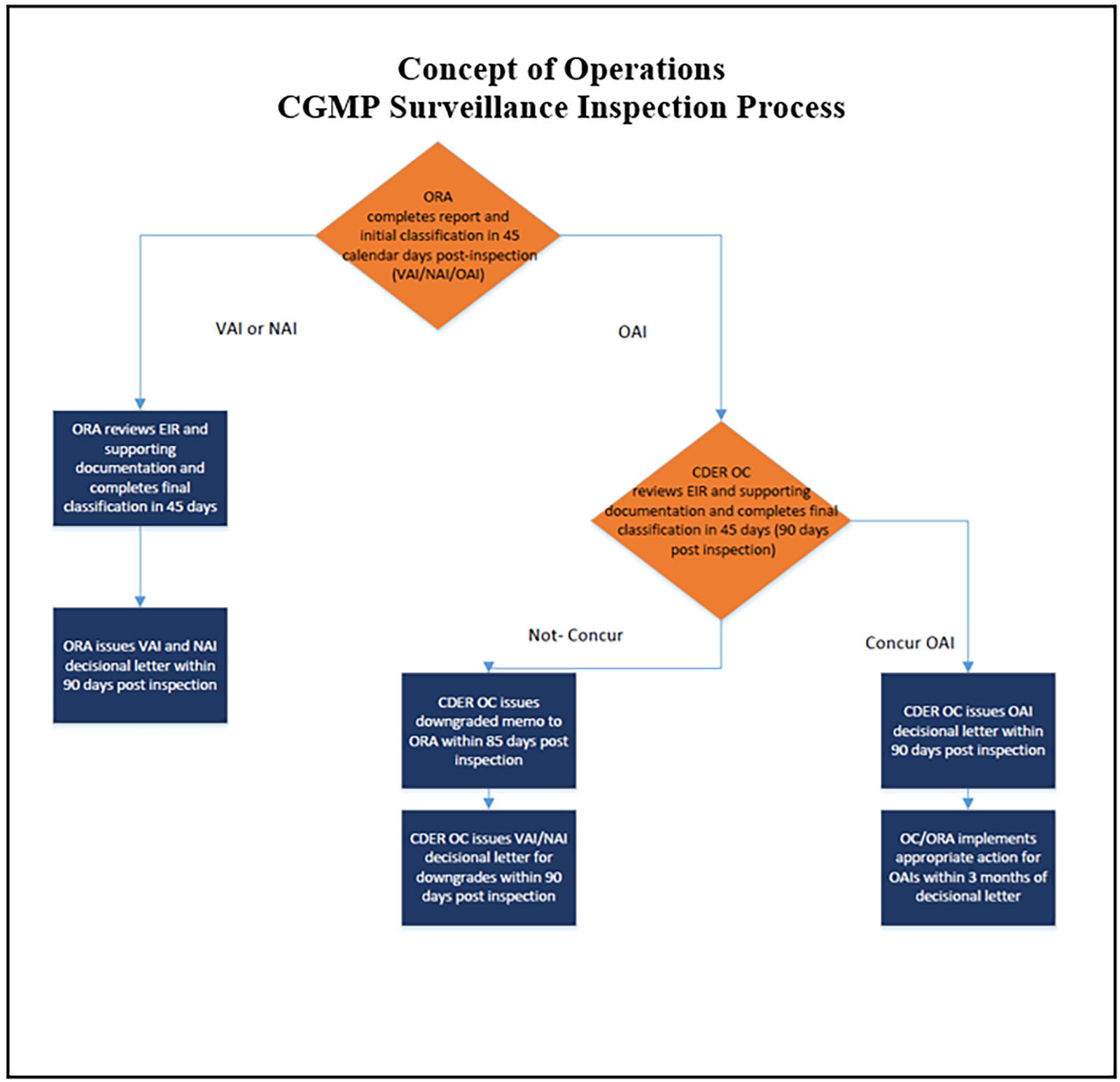
As part of ConOps the decision classification workflow process was redesigned. (See Figure 7). Under the ConOps agreement, when ORA initially recommends classifying the inspection as OAI, CDER reviews the report along with any remediation plan or response submitted by the company. CDER evaluates the evidence supporting inspection observations, potential impact to patient safety, the company’s responses to the observations, and the adequacy of proposed corrective actions. CDER may reclassify the inspection based on this review. CDER also can, and has, upgraded classifications to OAI, even when initial recommendations from the field are for an acceptable classification. This typically occurs with for-cause inspections where the proposed corrective actions by the firm are determined by CDER to be inadequate.
Figure 7. Process for classifying surveillance inspection outcomes after implementation of the ConOps.
Implementation of the ConOps has improved consistency in evaluation of inspection observations and classifications and has reduced the time frames for taking enforcement action. The percentage of cases in which CDER concurs with ORA’s initial recommendation is known as the “concurrence rate” (See Figure 8). In 2019, the concurrence rate had risen to 73 percent.
Figure 8. Concurrence rates on foreign drug inspections designated OAI were 50% in 1996 and rose to 73% in 2019. (FY 1996-1997 based on GAO data, all other data from FDA compliance database.)
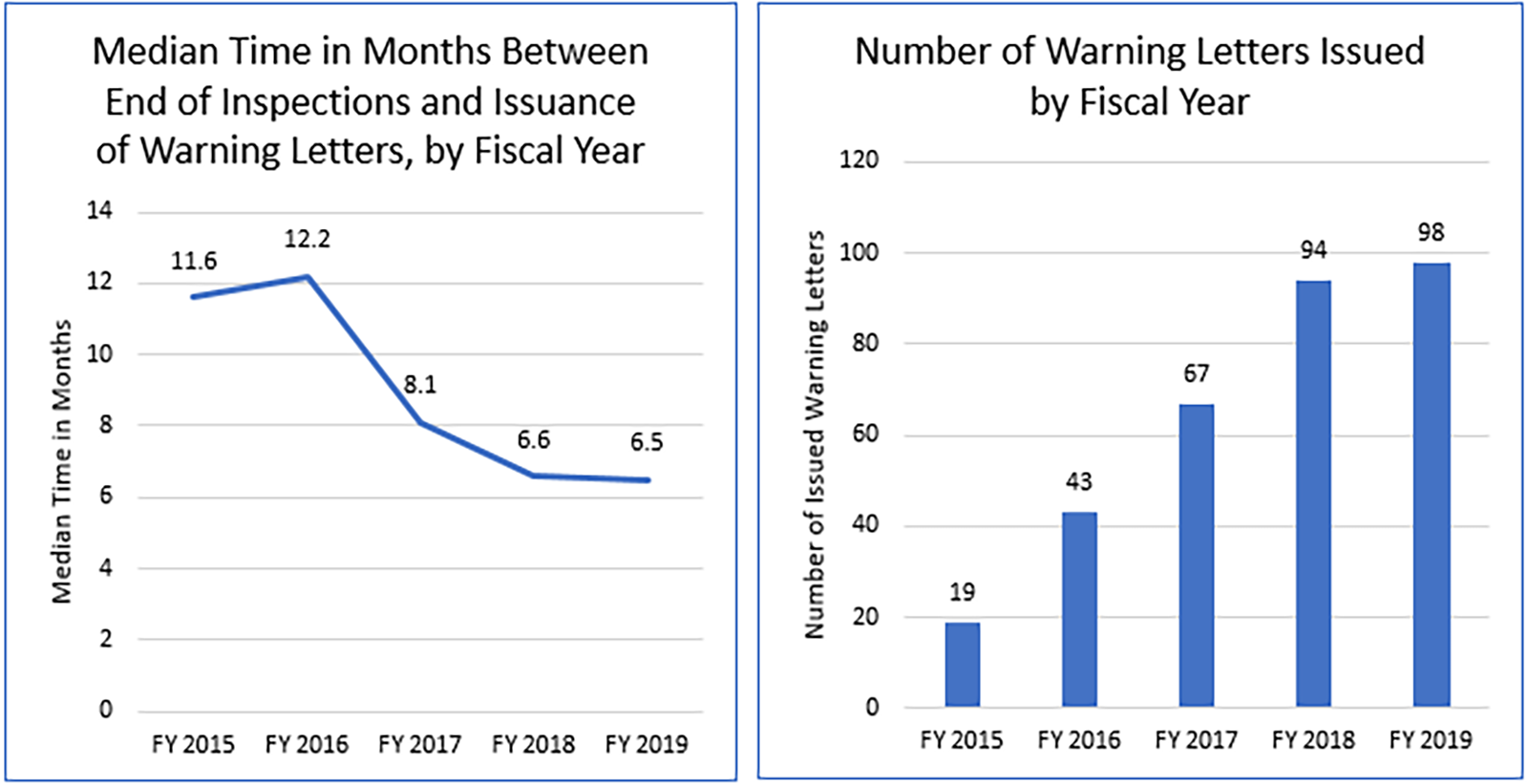
The median time for FDA to issue a warning letter for drug manufacturing issues has decreased since ConOps was implemented, even though the number of warning letters FDA has issued has increased during that same time period (See Figure 9).
Agency Progress Toward Six-Month Compliance Actions
Figure 9. From FY 2015 to FY 2019 there has been an overall median 44% improvement in median time between the end of an inspection and issuance of a warning letter. During the same time, the number of warning letters increased.
Building an Investigator Work Force
Since 2015, FDA has performed more foreign than domestic inspections. The Agency has done so by using a mixed investigator work force consisting of (1) U.S.-based investigators who perform both domestic and foreign inspections; (2) a dedicated foreign cadre of U.S.-based drug investigators who conduct foreign inspections exclusively; and (3) foreign office-based experienced investigators who inspect facilities manufacturing human drugs (See Table 1). The majority of foreign inspections are performed by U.S.-based investigators.
Type of Investigator |
Number of Qualified Foreign Drug Investigators in FY 2019 | Number of Foreign Inspections Each Investigator is Expected to Perform Each Year | Estimated Percentage of All Foreign Inspections Performed in FY 2019 |
|---|---|---|---|
| U.S.-Based Investigators Performing Foreign and Domestic Inspections | 188 | 3-6 Foreign inspections per year | 90% |
| Dedicated Foreign Drug Cadre Based in US | 11 (included in the 188 listed above) | 16-18 inspections per year | 16% (part of the 90% above) |
| Foreign Office-Based Investigators | 10 | 15 inspections per year | 10% |
Table 1. FDA’s Investigator Work Force for Inspections of Foreign Facilities Producing Human Drugs, FY 2019
During calendar year 2019 ORA successfully hired and on-boarded 24 pharmaceutical investigators. In 2020 our investigator hiring efforts are continuing, and with our new direct hire authority we anticipate filling all our pharmaceutical investigator vacancies in 2020. In recent years, the Office of Global Policy and Strategy, which oversees FDA’s foreign offices, has made progress in developing the foreign office-based inspectorate. At the same time, FDA’s participation in the Mutual Recognition Agreement with the European Union has enabled us to focus more of our investigator work force on higher-risk facilities around the world.
The Agency continues to face challenges, however, in developing the investigator work force due to the rigorous nature of the job (e.g., foreign travel restrictions and hardship) and competition for qualified candidates. Once the Agency succeeds in hiring a new investigator, it can take 1.5 to 2 years of training to bring them to a fully proficient level. FDA also faces challenges to achieving optimum staffing levels, such as negotiated agreements with host countries that affect the number of investigators who can permanently attach to a foreign office.
COVID-19 and Inspection Impact
As noted at the beginning of this testimony, as a result of the COVID-19 pandemic, most foreign and domestic surveillance facility inspections are currently postponed. Only inspections deemed mission-critical will be considered on a case-by-case basis as this outbreak continues to unfold.
We employ additional tools to ensure the safety of products imported to the U.S., which have proved effective in the past. These include:
- Denying entry of unsafe products into the U.S.;
- Physical examinations and/or product sampling at our borders;
- Reviewing a firm’s previous compliance history;
- Using information sharing from foreign governments as part of mutual recognition and confidentiality agreements; and
- Requesting records “in advance of or in lieu of” on-site drug inspections.
Through our risk-based import screening tool (PREDICT), FDA has the ability to focus our examinations and sample collections based on heightened concerns of specific products being entered into U.S. commerce. The PREDICT screening continues to adjust risk scores as necessary throughout the COVID-19 outbreak.
FDA investigators remain on the front lines at ports of entry, quickly examining and reviewing import entries to help ensure goods being imported are consistent with FDA requirements and/or policies. We are in close communication with our partners at U.S. Customs and Border Protection to proactively identify and mitigate any potential backlogs.
FDA participates in FEMA Supply Chain Task Force meetings, providing regulatory support and subject matter expertise to respond to questions concerning medical products identified by FEMA, to facilitate the lawful entry and use of imported medical products coordinated through FEMA, and to inform medical product supply chain discussions.
FDA remains committed to using all available tools to oversee the safety and quality of FDA-regulated products for American patients and consumers. As this remains a dynamic situation, we will continue to assess and calibrate our approach as needed and we stand ready to resume any postponed inspections as soon as feasible.
FDA’s Sampling and Testing Program
Although application assessments and inspections are a foundation of FDA’s efforts to maintain a safe, reliable drug supply, the safety and effectiveness of drugs depends on a multipronged approach, of which quality checks by FDA and manufacturers are a part. To help ensure that safe and effective drugs are sold in the U.S., we test selected drugs in state-of-the-art FDA laboratories and through research contracts and grants. This testing program includes APIs and finished drug products. We test using the same standards that are part of the drug approval process for identity, strength, and purity.
Some have questioned why we do not test every drug product before it enters the U.S. FDA performs thousands of tests a year pre- and post-market. Only a small percentage (about one percent) of drugs that are tested fail to meet the established quality specifications. 11 Testing by FDA or third parties of each batch of drug product in U.S. commerce, which amounts to millions of batches and trillions of individual tablets, capsules, and other dosage forms, before they enter the U.S. market would not be feasible at a practical level (in 2018, there were almost 186 trillion tablets and capsules on the U.S. market 12) and the current approach is effective and efficient.
Additional Drug Safety and Surveillance Efforts
Ongoing review and surveillance efforts can identify new safety concerns that require quick action. When they do, the Agency makes every effort to investigate potential health risks and provide our recommendations to the public based on the best available science.
As an example, in April, FDA requested that manufacturers withdraw all prescription and over-the-counter (OTC) ranitidine drugs from the market immediately. This was the latest step in an ongoing investigation of a contaminant known as N-Nitrosodimethylamine (NDMA) in ranitidine medications (one commonly known brand name is Zantac). FDA began an investigation into potential NDMA contamination in drug products containing ranitidine when it first obtained information that there was a possibility of impurities in those products. NDMA is a probable human carcinogen (a substance that could cause cancer).
Last summer, the Agency became aware of independent laboratory testing that found NDMA in ranitidine. Low levels of NDMA are commonly ingested in the diet; for example, NDMA is present in foods and in water. These low levels would not be expected to lead to an increase in the risk of cancer. However, sustained higher levels of exposure may increase the risk of cancer in humans. The Agency conducted thorough laboratory tests and found NDMA in ranitidine at low levels. At the time, the Agency did not have enough scientific evidence to recommend whether individuals should continue or stop taking ranitidine medicines. FDA continued its investigation and warned the public last fall of the potential risks and to consider alternative OTC and prescription treatments.
New FDA testing and evaluation confirmed that NDMA levels increase in ranitidine even under normal storage conditions, and NDMA has been found to increase significantly in samples stored at higher temperatures, including temperatures the product may be exposed to during distribution and handling by consumers. The testing also showed that the older a ranitidine batch is, or the longer the length of time since it was manufactured, the greater the level of NDMA, possibly resulting in ranitidine product being above the acceptable daily intake limit.
Based on this information, FDA took swift action to assure that ranitidine products will no longer be available for new or existing prescriptions or OTC use in the U.S.
FDA Encourages Industry to Invest in Mature Quality Management Systems and Advanced Manufacturing Technology
FDA inspects manufacturing facilities and takes action, if needed, to enforce CGMP quality standards and applicable regulations. The Agency’s investigators look for deficiencies in meeting CGMP standards, but these assessments do not measure how far the facility is above the minimum CGMP. Simple adherence to CGMP standards does not indicate that a firm is investing in improvements or planning or deploying advanced quality control techniques that could better enable it to prevent quality problems leading to supply disruptions.
This is why it is critical that industry evolve from meeting the minimum manufacturing quality threshold to achieving quality management maturity. Some pharmaceutical firms have been slow to implement robust, mature quality systems and the accompanying quantitative measures of quality that have been the foundation of success in other industries, such as automotive and aerospace. 13 These industries exercise quality oversight by continuously monitoring quality in real time during manufacturing of their products, and promptly correcting operations when needed. Numerous organizations and quality experts have worked to develop conceptual models and standards for advancing the maturity of industrial quality management systems. These models could be used more broadly in the pharmaceutical industry to improve the quality and reliability of the drug supply.
Many pharmaceutical manufacturers, whether domestic or foreign, have been slow to invest in these mature quality management systems because the market currently has no visibility into manufacturing facilities’ quality. This lack of transparency reinforces competition based solely on price and disincentivizes companies from making investments in upgrading their facilities and quality practices until problems become frequent and severe enough to result in supply disruptions and drug shortages. As we have stated in our recent report, “Drug Shortages: Root Causes and Potential Solutions”, 14 a way to create incentives for manufacturers to invest in product quality is to develop and implement a rating system for quality management maturity that is based on objective criteria. Such a rating system could enable purchasers to compare differences in quality and choose whether to reward more reliable manufacturers financially and with increased market share.
In addition to quality management maturity, the Agency encourages pharmaceutical manufacturers to invest in advanced manufacturing technology to improve their products and processes. Although widely used in some other industries, such as automotive, aerospace, and semiconductors, advanced manufacturing is now just beginning to be used by pharmaceutical companies. New technologies include “continuous manufacturing” (CM), wherein the finished drug product or active pharmaceutical ingredient is produced as a continuous stream, as opposed to traditional batch manufacturing where breaks or stops exist between different processing steps. In some examples of advanced pharmaceutical manufacturing, production can be continuous from chemical synthesis of the active ingredient through production of the tablets or other dosage forms. Product quality can be precisely controlled with modern automation and control systems and can be closely monitored during production by using highly sensitive analytical tools. Other examples of advanced manufacturing include 3D printing, isolator technology, miniaturization, and robotics.
Conclusion
Thank you for the opportunity to discuss FDA’s oversight of pharmaceutical manufacturing. COVID-19 has provided yet more proof that to protect the reliability and availability of drugs to treat Americans is of vital importance. We look forward to working with the Committee and others to strengthen investment in modern manufacturing technology, establish incentives for mature quality management systems, and consider additional measures.
We are happy to answer any questions.
Footnotes
1. Bumpas, Janet; Betsch, Ekkehard. Exploratory study on active pharmaceutical ingredient manufacturing for essential medicines (English). Health, Nutrition and Population (HNP) discussion paper. Washington, DC: World Bank: 12 – 13, Figure 2. http://documents.worldbank.org/curated/en/848191468149087035/Exploratory-study-on-active-pharmaceutical-ingredient-manufacturing-for-essential-medicines. Accessed September 30, 2019.
2. U.S. Food and Drug Administration, “Pathway to Global Product Safety and Quality,” A Special Report, p. 20. Accessed October 4, 2019 at https://www.hsdl.org/?view&did=4123.
3. The Agency updates the Catalog continually, so the information it provides is a snapshot in time.
4. The Federal Food, Drug, and Cosmetic Act (FD&C Act) describes different circumstances in which a drug may be considered adulterated. For example, a drug might be adulterated where it is contaminated with filth, where its purity departs from certain compendial standards, or where the conditions of its manufacturing are not consistent with current good manufacturing practice (CGMP).
5. The 2016 GAO report identifies 965 firms that, at that time, had not been inspected. Since then, there were two separate mergers of facilities in that count, dropping the number from 965 to 963.
6. Under the FD&C Act, as amended by FDASIA, a drug product will be deemed adulterated if it has been manufactured, processed, packed, or held in any factory, warehouse, or establishment which delays, denies, or limits an inspection, or refuses to permit entry or inspection. In such a case, FDA typically will place the firm on import alert.
7. See “What Is A Classification?” at https://www.fda.gov/inspections-compliance-enforcement-and-criminal-investigations/inspection-references/inspections-database-frequently-asked-questions.
8. Import Alert: Import alerts inform the FDA's field staff and the public that the agency has enough evidence to allow for Detention Without Physical Examination (DWPE) of products that appear to be in violation of the FDA's laws and regulations. These violations could be related to the product, manufacturer, shipper and/or other information.
11. These are established in approved applications, and for many drugs also by USP (https://qualitymatters.usp.org/what-usp-standard).
12. IQVIA. National Sales Perspective. 2014-2018. Extracted: August 2019.
13. Fuhr, Ted, et al., 2015, Flawless-from Measuring Failure to Building Quality Robustness in Pharma, McKinsey & Company.