remedē® System – P160039
This is a brief overview of information related to FDA’s approval to market this product. See the links below to the Summary of Safety and Effectiveness Data (SSED) and product labeling for more complete information on this product, its indications for use, and the basis for FDA’s approval.
Product Name: remedē® System
PMA Applicant: Respicardia, Inc.
Address: 12400 Whitewater Drive, Suite 150 Minnetonka, MN 55343
Approval Date: October 6, 2017
Approval Letter: central sleep apnea in adults.
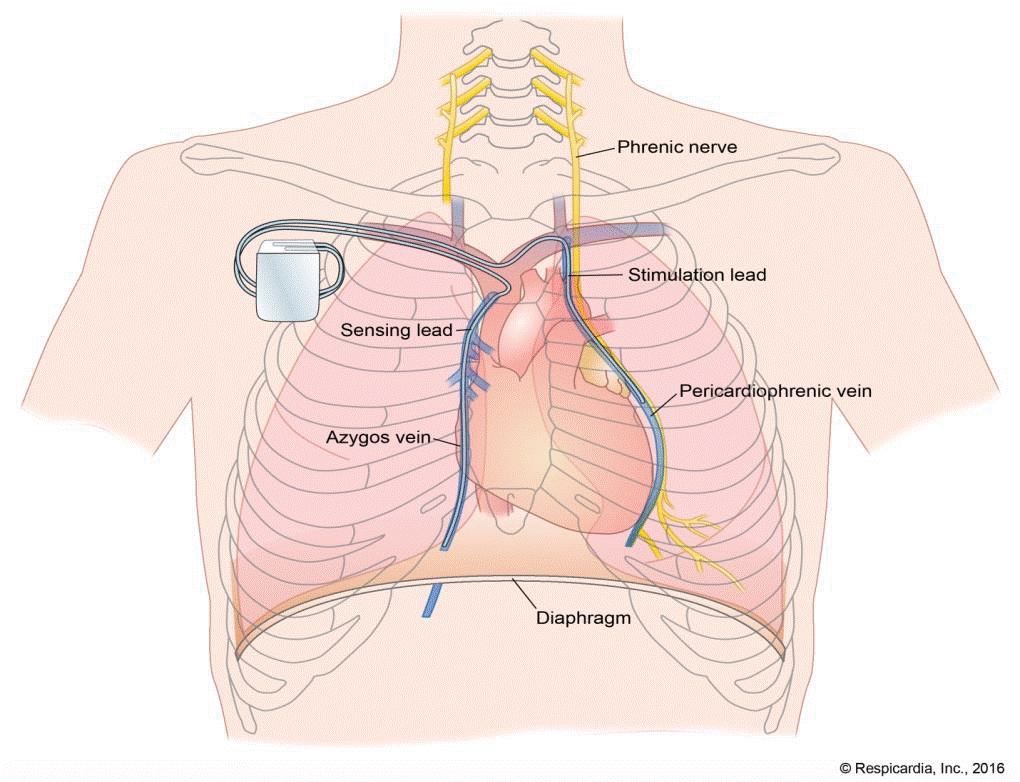
What is it? The remedē System is an implanted nerve stimulator used to treat moderate to severe central sleep apnea (CSA) in adults. The system includes a battery-powered pulse generator that is implanted under the skin in the upper chest and thin wire leads that are threaded through veins near the nerve that stimulates breathing (phrenic nerve). The system is programmed using an external System Programmer and Programming Wand.
How does it work? The remedē System delivers a small electrical stimulus to the phrenic nerve while a patient is asleep. This stimulus makes the diaphragm muscle contract, which causes the patient to take a breath. The remedē System has 2 modes – it can be set to generate pulses at a fixed rate (asynchronous therapy) or it can deliver a pulse only when it detects a pause in breathing (synchronous therapy). The physician is able to set the stimulator to deliver the most appropriate therapy for the patient. The system has safeguards to make sure that therapy is only delivered during sleep, for example it works only at the time of day when the patient is expected to be sleeping, and it turns on only when the patient is inactive and lying down.
When is it used? The device is used for the treatment of moderate to severe
What will it accomplish? The clinical trial used to support the approval of this system included 151 patients. The study measured the apnea hypopnea index (AHI) in adult patients with moderate to severe central sleep apnea. This index is a measure of the frequency and severity of apnea episodes during sleep. The study demonstrated significant reduction in the AHI in many patients using the remedē System compared to a control group which did not receive the device therapy. After six (6) months, the AHI was reduced by at least half in 51 percent of patients who had an active remedē System implanted compared to only 11 percent of patients in a control group. The most common adverse events reported included interactions with other implanted devices, movement or dislodgement of the leads, discomfort, implant site infection, swelling, and local tissue damage (pocket erosion).
When should it not be used? The remedē System should not be used in patients who have an active infection or in patients known to need magnetic resonance imaging (MRI).
Additional information (including warnings, precautions, and adverse events): Summary of Safety and Effectiveness Data and labeling are available at:
Other Resources: Press Release