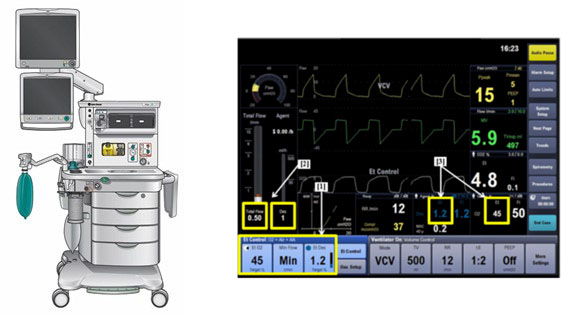
Et Control - P210018
This is a brief overview of information related to FDA’s approval to market this product. See the links below to the Summary of Safety and Effectiveness Data (SSED) and product labeling for more complete information on this product, its indications for use, and the basis for FDA’s approval.
Product Name: Et Control
Applicant: Datex-Ohmeda, Inc., a General Electric Company
Address: Datex-Ohmeda, Inc., 3030 Ohmeda Drive, PO Box 7550, Madison, WI 53707-7550 USA
Approval Date: March 17, 2022
Approval Letter: Approval order
What is it?
Et Control is a software feature that is indicated for use with the Datex-Ohmeda Aisys CS2 anesthesia system. When a patient is under anesthesia, the software helps patients reach and maintain target exhaled oxygen levels (targeted end tidal levels) and the right mixture of anesthetic gas to control pain (end tidal anesthetic) that are set by the health care provider.
How does it work?
Et Control, an optional feature, allows health care provider to set target end tidal oxygen (EtO2) and end tidal anesthetic agent (EtAA) levels on the Aisys CS2 host anesthesia machine. The Et Control software takes breath-by-breath measurements and works with the anesthesia machine. It continuously measures and adjusts (titrates) the electronic gas mixer and anesthetic agent vaporizer to reach and maintain target levels.
When is it used?
The Et Control feature is indicated for use with patients 18 years of age and older. It is designed to work together with the Aisys CS2 Anesthesia System to maintain oxygen and anesthesia gas concentrations that are set by the health care provider for a procedure with anesthesia.
What will it accomplish?
The Et Control feature helps the health care provider maintain low flow anesthesia, decreases anesthetic agent use and greenhouse gas emissions, and reduces the workload needed to accomplish these goals.
When should it not be used?
There are no known reasons not to use this device.