Chocolate Touch Paclitaxel Coated PTA Ballon Catheter - P210039
This is a brief overview of information related to FDA’s approval to market this product. See the links below to the Summary of Safety and Effectiveness Data (SSED) and product labeling for more complete information on this product, its indications for use, and the basis for FDA’s approval.
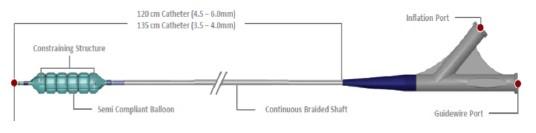
Product Name: Chocolate Touch Paclitaxel Coated PTA Balloon Catheter
PMA Applicant: TriReme Medical, LLC
Address: 7060 Koll Center Parkway, Suite #300, Pleasanton, CA 94566
Approval Date: November 4, 2022
Approval Letter: Approval order
What is it?
The Chocolate Touch Paclitaxel-coated PTA Balloon Catheter uses a drug-coated balloon (DCB) to re-open blocked or narrowed arteries in the thigh and knee due to peripheral artery disease (PAD). The balloon is coated on its outer surface with the drug paclitaxel; a drug which may help prevent the arteries from narrowing again (restenosis).
How does it work?
A traditional angioplasty balloon catheter is inserted through the blood vessels, across the blockage or narrowing, and inflated to partially open the blockage or narrowing. The Chocolate Touch DCB is then used to fully open the narrowed portion of the artery and apply the drug to the artery wall.
When is it used?
The Chocolate Touch DCB is used when arteries in the thigh and knee are narrowed or blocked as a result of peripheral artery disease. PAD occurs when fatty material (plaque) builds up in these arteries. The buildup can cause hardening and/or narrowing of the arteries (atherosclerosis), limiting the flow of oxygen-rich blood to the body. People with PAD may experience lifestyle-limiting symptoms, such as leg pain, or serious complications, including skin ulcers or gangrene.
What will it accomplish?
The Chocolate Touch DCB opens up a narrow or blocked artery in the thigh or knee. It was shown to be as safe and effective as other drug coated balloons. At 12 months post-procedure, the Chocolate Touch DCB was able to keep 77.1% of arteries open.
When should it not be used?
The Chocolate Touch DCB should not be used in:
- Arteries that carry blood to the heart (coronary), kidneys (renal), brain (cerebrovascular), or branch off from the largest artery (supra-aortic).
- Patients with a known hypersensitivity (allergy) to paclitaxel or drugs with similar characteristics as paclitaxel.
- Patients who cannot take recommended medicines that thin the blood and prevent blood clots.
- Women who are breastfeeding, pregnant, or intend to become pregnant; or men intending to father children.
- If a doctor decides that a patient’s narrowing or blockage will prevent proper placement of the delivery system.