Medtronic HeartWare Ventricular Assist Device (HVAD) System
Update: September 22, 2023
The FDA added information for health care providers about Medtronic’s September 2023 communication, which notified users that the issue with the HVAD System Autologs web portal is now resolved. Additional information can be found on the Recalls Related to HVAD System web page.
Update: August 25, 2023
Please see Medtronic's August 23, 2023, Urgent Medical Device Communication for updated patient management recommendations for HeartWare Ventricular Assist Device (HVAD) systems. The FDA agrees with Medtronic's recommendations and references select recommendations on the Recommendations for Patients and Health Care Providers: HVAD System page and on the Recalls Related to HVAD System page.
While no new patients have received the HVAD system since Medtronic stopped the sale and distribution in June 2021, we are sharing this new information that impacts certain patients who already have HVAD systems.
- Medtronic's Urgent Medical Device communication describes a newly identified subgroup of HVAD pumps defined by serial numbers (subgroup 3) that have a higher risk of failure to restart than the general population of patients with HVAD pumps.
- Patients in a subgroup should contact their VAD coordinator if they need to make a controller exchange so that the exchange can take place in a clinical setting.
Medtronic stopped the sale and distribution of the HeartWare Ventricular Assist Device (HVAD) system on June 3, 2021, given the increased risk of mortality and neurological adverse events in patients using the device, and a malfunction where the device may fail to restart. Both problems may lead to serious injuries or death.
The FDA continues to work with Medtronic to ensure the health and safety of device users, which remains our highest priority. This includes ensuring Medtronic continues to meet its obligations to support patients and health care providers.
The FDA is committed to providing important updates about the HeartWare device and we will continue to inform the public as new information becomes available.
Information about the HVAD System
Recommendations for Patients and Health Care Providers
Recalls Related to HVAD System
On this page:
What is the HVAD System?
Heart failure is a chronic condition that typically worsens over time and can result in end-stage heart failure and death. Patients with end-stage heart failure suffer debilitating shortness of breath, physical exhaustion, painful swelling in legs and feet, and fluid buildup in the lungs and abdomen.
The Medtronic HVAD System helps the heart by supporting the weak left ventricle and providing additional blood flow. The Medtronic HVAD System is a ventricular assist device used in end-stage heart failure patients who are waiting for a heart transplant. This use is known as bridge-to-transplant. The Medtronic HVAD System may also be used in end-stage heart failure patients who cannot have a heart transplant, and whose symptoms cannot be managed by medications. This use is known as destination therapy, which can extend a patient's life and improve their quality of life.
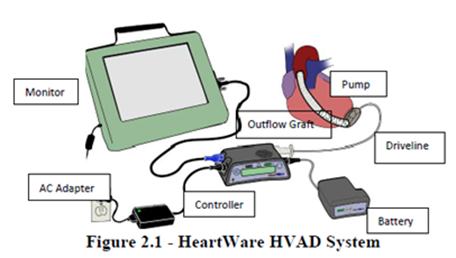
The Medtronic HVAD System includes a pump implanted in the space around the heart (pericardium), a controller that controls the speed and function of the pump, a tube inserted into the heart to drain the blood, and a tube inserted in the aorta for the blood that is being pumped. The controller remains outside the patient's body and is connected to the pump by a flexible tube that passes through the patient's skin. The controller is powered either by batteries or a power supply that can be connected to a standard electrical power outlet.
Benefits and Risks of the HVAD System
As discussed in the Agency's benefit-risk assessments in 2012 and 2017, and for patients currently supported on the device, the probable benefits of the HVAD System outweigh the probable risks. The benefits of the HVAD system include survival on the device, ability to place the HVAD pump without requiring an abdominal pocket, and ability to place the device in smaller patients when compared to the alternatives. The probable risks include stroke and other neurological events, major infection, bleeding, and right heart failure, all of which could lead to death. The FDA recognizes neurological events, in particular strokes, associated with the HVAD System as the most important probable risk. The current labeling for the HVAD System for patients and health care providers conveys these benefits and risks.