Pharmaceutical Quality - Chemistry, Manufacturing & Controls | PQ/CMC
PQ/CMC Recent Highlights
Updates on PQ/CMC FHIR Implementation Guide Development
Please note that HL7 Connectathon testing, balloting, and publication of versions of a FHIR Implementation Guide (IG) do not represent the IG in its final form nor do these activities in any way constitute a statement of policy or guidance from FDA. There are formal processes and timelines by which FDA announces support for accepting data in a particular format.
Until such time, the growing PQ/CMC HL7 FHIR Implementation Guide is available to review at HL7.org. As new updates are added to the IG at HL7, there will be opportunities to participate in testing new IG content at HL7 Connectathons and to comment during balloting of the new content.
The PQ/CMC and HL7 FHIR page has been significantly updated to reflect the stages of PQ/CMC FHIR IG development, status, and relevant links to HL7 IG pages.
- The Stage 1 Implementation Guide (IG) for “Pharmaceutical Quality - Chemistry, Manufacturing and Controls (PQ-CMC) Submissions to FDA” has been published at HL7 as a Standard for Trial Use (STU) version 1.0.0.
- Stage 2 of the PQ/CMC FHIR Implementation Guide, adding support for new PQ/CMC data content, was successfully tested at the September 2024 HL7 FHIR Connectathon and is intended for inclusion in the January 2025 HL7 Ballot cycle. The link for the Stage 2 IG being presented for ballot can be found at https://hl7.org/fhir/us/pq-cmc-fda/2025Jan/.
Overview of the PQ/CMC Effort
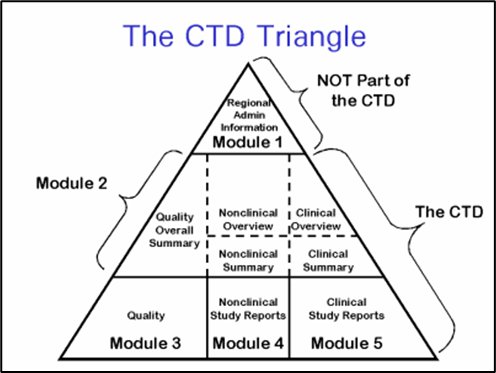
As part of an effort intended to support future electronic acquisition and use of submitted information, FDA has undertaken a project to identify and prioritize pharmaceutical quality/chemistry, manufacturing and controls (PQ/CMC) information that would benefit from a structured submission approach. This structured and standardized information is intended to be submitted in the Module 3 of the Common Technical Document as defined by the International Council for Harmonisation’s (ICH) M4 Common Technical Document (CTD). The effort is scoped to some of what is currently submitted in Module 3 and Module 2.3 of the electronic Common Technical Document (eCTD) submission. It is not intended to be comprehensive in structuring all eCTD product quality information, only those concepts that were considered amenable to structuring and would bring value to the quality review process.
The goal of this project is to establish electronic standards for submitting Pharmaceutical Quality (PQ) and Chemistry & Manufacturing Controls (CMC) data.
Specific objectives of the PQ/CMC Project are:
- Develop structured data standards for PQ/CMC
- Develop a data exchange standard for submitting the structured PQ/CMC data to the FDA
The submission of structured data in a standardized format should increase the efficiency of FDA’s review of PQ/CMC data contained in the Module 3 of eCTD submissions for a New Drug Application (NDA), an Investigational New Drug Application (IND), a Biologics License Application (BLA), an Abbreviated New Drug Application (ANDA), a New Animal Drug Application (NADA), an Abbreviated New Animal Drug Application (ANADA), an Investigational New Animal Drug (INAD), Generic Investigational New Animal Drugs (JINADs), and a Master File (MF).
For consistency of product quality data across FDA centers, the draft standardized data elements and terminologies were created by an Agency workgroup comprised of Subject Matter Experts (SMEs) from the Center for Drug Evaluation and Research (CDER), the Center for Veterinary Medicine (CVM), and the Center for Biologics Evaluation and Research (CBER).
The PQ/CMC Data exchange standard is intended to be developed as Health Level 7 (HL7) Fast Health Interoperable Resources (FHIR) representation. The project has been established with the HL7 Biomedical Research & Regulation (BR&R) work group and discussions regarding representing the PQ/CMC structured elements in FHIR occur during the work group meetings. These HL7 BR&R WG meetings are open to the public.
Figure 1: ICH CTD Modules
Stay Connected
- For additional information/support from PQ/CMC Team, please contact PQ-CMC@fda.hhs.gov.