Aspartame and Other Sweeteners in Food
FDA Response to External Safety Reviews of Aspartame
The FDA is aware of the International Agency for Research on Cancer (IARC) and Joint FAO/WHO Expert Committee on Food Additives (JECFA) conclusions about aspartame issued July 14, 2023. Aspartame being labeled by IARC as “possibly carcinogenic to humans” does not mean that aspartame is actually linked to cancer.
The FDA disagrees with IARC’s conclusion that these studies support classifying aspartame as a possible carcinogen to humans. FDA scientists reviewed the scientific information included in IARC’s review in 2021 when it was first made available and identified significant shortcomings in the studies on which IARC relied. We note that JECFA did not raise safety concerns for aspartame under the current levels of use and did not change the Acceptable Daily Intake (ADI).
Aspartame is one of the most studied food additives in the human food supply. FDA scientists do not have safety concerns when aspartame is used under the approved conditions. The sweetener is approved in many countries. Regulatory and scientific authorities, such as Health Canada and the European Food Safety Authority have evaluated aspartame and also consider it safe at current permitted use levels.
Some consumers may rely on products with aspartame and other sweeteners to help reduce their sugar consumption. We recognize that navigating different information from health organizations is challenging. We will continue to provide reliable, science-based information on aspartame and other sweeteners on the FDA’s web site to help consumers make informed choices.
Sweeteners or sugar substitutes, such as aspartame, sucralose, and stevia derived substances, are ingredients used to sweeten and in some cases enhance the flavor of foods. Some sweeteners are much sweeter than table sugar, and smaller amounts are needed to achieve the same level of sweetness as sugar in food. People may choose to use sweeteners instead of sugar for various reasons. For example, sweeteners contribute only a few or no calories to the diet and generally will not raise blood sugar levels.
Sweeteners are widely used in foods and beverages marketed as "sugar-free" or "diet," including baked goods, soft drinks, powdered drink mixes, candy, puddings, canned foods, jams and jellies, dairy products, and many other foods and beverages. Consumers can identify whether a product has a sweetener by looking for the sweetener’s name in the ingredient list on the product label.
By law, like all other ingredients added to food in the U.S., sweeteners must be safe for consumption. Companies are responsible for ensuring the regulatory status and safety of the ingredients in their products before marketing. Under its legal authorities, the FDA administers pre-market regulatory programs that assist companies in meeting their obligations. To market a new food additive or before using a food additive in a different way than how the FDA has currently approved it, a manufacturer or other sponsor must first seek approval from the FDA by submitting a food additive petition. Under the law, certain ingredients do not require pre-market food additive approval by FDA, for example, if they are generally recognized as safe (GRAS) by qualified experts. If a company concludes that the specific use of a sweetener is GRAS, they may submit their information to the FDA through the FDA’s GRAS Notification Program. Information about the regulatory status of sweeteners is available in the Code of Federal Regulations and the FDA’s public GRAS Notice Inventory and Substances Added to Food (formerly EAFUS) Inventory.
The FDA continues to monitor the latest science available on sweeteners in a variety of ways. FDA scientists reassess the science about the exposure and safety of a sweetener each time the agency files a food additive petition or a GRAS notice for that sweetener. We also stay abreast of published literature and the current level of consumer exposure and participate in international scientific and standard-setting activities related to food ingredient safety.
Sweeteners Authorized as Food Additives in the U.S.
Based on the available scientific evidence, sweeteners authorized by the FDA are safe for the general population under certain conditions of use. There are food additive listings for six sweeteners, including:
Aspartame is approved for use in food as a sweetener. Aspartame brand names include Nutrasweet®, Equal®, and Sugar Twin®. It contains calories, but consumers will likely use less than they would of table sugar because it is about 200 times sweeter.
Aspartame [L-aspartyl-L-phenylalanine methyl ester] is a dipeptide composed primarily of two amino acids, phenylalanine, and aspartic acid. These, and other amino acids, are natural constituents of protein-containing foods consumed in any healthful diet. When phenylalanine and aspartic acid are combined in a certain way to form aspartame, they produce an intensely sweet-tasting substance. Aspartame is not heat stable and loses its sweetness when heated, so it typically isn’t used in baked goods.
The FDA regulates aspartame as a food additive. The FDA first issued a regulation for aspartame in 1974 for use as a tabletop sweetener and in chewing gum, cold breakfast cereals, and dry bases for certain foods (for example, beverages, instant coffee and tea, gelatins, puddings and fillings, and dairy products and toppings). Since that time, the FDA approved aspartame for other uses, including most recently as a general-purpose sweetener in 1996. For more information about the regulatory history of aspartame, see the Timeline of Selected FDA Activities and Significant Events Addressing Aspartame.
Scientific evidence has continued to support the FDA’s conclusion that aspartame is safe for the general population when made under good manufacturing practices and used under the approved conditions of use. The FDA-established acceptable daily intake (ADI), or the amount of aspartame that is considered safe to consume each day over the course of a person’s lifetime, continues to be protective of public health. However, people with difficulty metabolizing phenylalanine because of a rare genetic disorder called phenylketonuria (PKU) should avoid or restrict aspartame. Newborns are routinely tested for PKU using a “heel-prick” test before they leave the hospital. Consumers can identify whether a product has aspartame by looking for it by name in the ingredient list on the product label, which must include a statement to inform people with PKU that the product contains phenylalanine.
Aspartame is one of the most studied food additives in the human food supply. To determine the safety of aspartame, the FDA has reviewed more than 100 studies designed to identify possible toxic effects, including studies that assess effects on the reproductive and nervous systems, carcinogenicity, and metabolism. FDA scientists reassessed the science on the exposure and safety of aspartame in food each time the agency filed a food additive petition, received relevant information from other regulatory agencies and research institutions that have evaluated aspartame, and responded to objections that were raised related to the approved uses of aspartame. Since the last approved use in 1996, the FDA has continued monitoring the scientific literature for new information on aspartame. We stay abreast of published literature and the current level of consumer exposure and participate in international scientific and standard-setting activities related to food ingredient safety. Other regulatory agencies have evaluated aspartame, and it is approved in many countries.
Acesulfame potassium is approved for use in food as a sweetener. It is included in the ingredient list on the food label as acesulfame K, acesulfame potassium, or Ace-K. Acesulfame potassium is sold under the brand names Sunett® and Sweet One®. It is about 200 times sweeter than table sugar and is often combined with other sweeteners.
The FDA regulates acesulfame potassium (Ace-K) as a food additive. The FDA approved acesulfame potassium for use in specific food and beverage categories in 1988 and in 2003 approved it as a general-purpose sweetener and flavor enhancer in food, except in meat and poultry, under certain conditions of use. It is heat stable, meaning it stays sweet even when used at hot temperatures during baking, making it suitable as a sugar substitute in baked goods.
To determine the safety of acesulfame potassium, the FDA reviewed more than 90 studies designed to identify possible toxic effects, including studies on reproductive effects, carcinogenicity, and metabolism.
Sucralose is approved for use in food as a sweetener. Sucralose is sold under the brand name Splenda®. Sucralose is about 600 times sweeter than table sugar.
The FDA regulates sucralose as a food additive. The FDA approved sucralose for use in 15 food categories in 1998 and for use as a general-purpose sweetener for foods in 1999, under certain conditions of use. Sucralose is a general-purpose sweetener found in various foods, including baked goods, beverages, chewing gum, gelatins, and frozen dairy desserts. It is heat stable, meaning it stays sweet even when used at high temperatures during baking, making it suitable as a sugar substitute in baked goods.
To determine the safety of sucralose, the FDA reviewed more than 110 studies designed to identify possible toxic effects, including studies on the reproductive and nervous systems, carcinogenicity, and metabolism. The FDA also reviewed human clinical trials to address metabolism and effects on patients with diabetes.
Neotame is approved for use in food as a sweetener. Neotame is sold under the brand name Newtame® and is approximately 7,000 to 13,000 times sweeter than table sugar.
The FDA regulates neotame as a food additive. The FDA approved neotame for use as a general-purpose sweetener and flavor enhancer in foods (except in meat and poultry), under certain conditions of use, in 2002. It is heat stable, meaning it stays sweet even when used at high temperatures during baking, making it suitable as a sugar substitute in baked goods.
To determine the safety of neotame, the FDA reviewed data from more than 110 animal and human studies designed to identify possible toxic effects, including effects on the immune, reproductive, and nervous systems.
Advantame is approved for use in food as a sweetener. It is approximately 20,000 times sweeter than table sugar (sucrose).
The FDA regulates advantame as a food additive. The FDA approved advantame for use as a general-purpose sweetener and flavor enhancer in foods (except in meat and poultry), under certain conditions of use, in 2014. It is heat stable, meaning it stays sweet even when used at high temperatures during baking, making it suitable as a sugar substitute in baked goods.
To determine the safety of advantame, the FDA reviewed data from 37 animal and human studies designed to identify possible toxic effects, including effects on the immune system, reproductive and developmental systems, and nervous system. The FDA also reviewed pharmacokinetic and carcinogenicity studies, and several additional exploratory and screening studies.
Saccharin brand names include Sweet and Low®, Sweet Twin®, Sweet'N Low®, and Necta Sweet®. It is 200 to 700 times sweeter than table sugar (sucrose) and contains no calories.
The FDA has regulated saccharin as a food additive since 1977. First discovered and used in 1879, saccharin is currently approved for use, under certain conditions, in beverages, fruit juice drinks, and bases or mixes when prepared for consumption per directions, as a sugar substitute for cooking or table use, and in processed foods. Saccharin is also listed for use for certain technological purposes.
In the early 1970s, saccharin was linked with the development of bladder cancer in laboratory rats. This link led Congress to mandate additional studies of saccharin and the presence of a warning label on saccharin-containing products until such a warning could be considered unnecessary. Since then, more than 30 human studies demonstrated that the results found in rats were irrelevant to humans and that saccharin is safe for human consumption. In 2000, the National Toxicology Program of the National Institutes of Health concluded that saccharin should be removed from the list of potential carcinogens. Products containing saccharin no longer have to carry the warning label.
Plant and Fruit Based Sweeteners
In addition to the six sweeteners listed as food additives, the agency evaluated GRAS notices and has no questions about the GRAS conclusions for three types of plant and fruit-based high-intensity sweeteners:
Steviol glycosides are natural constituents of the leaves of Stevia rebaudiana (Bertoni) Bertoni, a plant native to parts of South America and commonly known as Stevia. They are reported to be 200 to 400 times sweeter than table sugar. They are used as general-purpose sweeteners in foods.
The FDA has evaluated many GRAS notices for the use of high purity (95% minimum purity) steviol glycosides, including Rebaudioside A (also known as Reb A), Stevioside, Rebaudioside D, or steviol glycoside mixture preparations with Rebaudioside A and/or Stevioside as predominant components. FDA has not questioned the notifiers' GRAS conclusions for these high-purity stevia derived sweeteners under the intended conditions of use identified in the GRAS notices submitted to the FDA. The FDA's response letters on such high purity steviol glycosides are available at the FDA's GRAS Notice Inventory website.
The use of stevia leaf and crude stevia extracts is not considered GRAS, and their import into the U.S. is not permitted for use as sweeteners. For details, see Import Alert 45-06.
The safety of steviol glycosides has been extensively studied and reported in the scientific literature. In humans, steviol glycosides are not hydrolyzed by digestive enzymes of the upper gastrointestinal tract and are not absorbed through the upper portion of the gastrointestinal tract. Several chronic studies and clinical studies in humans have been conducted demonstrating no adverse effects.
Siraitia grosvenorii Swingle fruit extract (SGFE) contains varying levels of mogrosides, which are the constituents of the fruit primarily responsible for the characteristic sweetness of SGFE. SGFE, depending on the mogroside content, is reported to be 100 to 250 times sweeter than sugar. Siraitia grosvenorii Swingle, commonly known as Luo Han Guo or monk fruit, is a plant native to Southern China. These fruit extracts are intended for use as general-purpose sweeteners and as tabletop sweeteners.
The FDA has evaluated GRAS Notices for SGFE. The FDA has not questioned the notifiers’ GRAS conclusions for SGFE under the intended conditions of use identified in the GRAS notices submitted to the FDA. The FDA’s response letters on SGFE are available on the FDA’s GRAS Notice Inventory website.
SGFE was the subject of a number of published studies including subchronic studies, clinical studies, and genotoxicity studies.
Thaumatin is a group of intensely sweet basic proteins isolated from the fruit of Thaumatococcus danielli (West African Katemfe fruit). Thaumatin is intended for use as a sweetener in various foods, including wine and other fermented or distilled beverages, jams, ice cream, bakery items, potato-based and similar snacks, and breakfast cereals. Thaumatin has a sweetness 2000-3000 times that of table sugar.
The FDA has evaluated several GRAS Notices for thaumatin and has not questioned the notifiers’ general recognition of safety conclusions for Thaumatin under the intended conditions of use identified in the notices submitted to the FDA. The FDA’s response letters on thaumatin are available at the agency’s GRAS Notice Inventory website.
Thaumatin has been the subject of numerous published sub-chronic studies as well as mutagenicity and allergenicity studies.
Sweetness Intensity of Sweeteners Compared to Table Sugar
The image below shows the sweetness intensity of sweeteners compared to table sugar or sucrose.
| Sweetener | Examples of Brand Names | Times Sweeter |
|---|---|---|
| Advantame | Advantame® | 20,000 times sweeter than table sugar (sucrose) |
| Neotame | Newtame® | 7,000 to 13,000 times sweeter than table sugar (sucrose) |
| Thaumatin | Talin® | 2,000 to 3,000 times sweeter than table sugar (sucrose) |
| Saccharin | Sweet and Low®, Sweet Twin®, Sweet'N Low®, Necta Sweet® | 200 to 700 times sweeter than table sugar (sucrose) |
| Sucralose | Splenda® | 600 times sweeter than table sugar (sucrose) |
| Steviol Glycosides | Truvia®, PureVia®, Enliten® | 200 to 400 times sweeter than table sugar (sucrose) |
| Luo Han Guo | Nectresse®, Monk Fruit in the Raw®, PureLo® | 100 to 250 times sweeter than table sugar (sucrose) |
| Aspartame | Nutrasweet®, Equal®, Sugar Twin® | 200 times sweeter than table sugar (sucrose) |
| Acesulfame Potassium (Ace-K) | Sweet One®, Sunett® | 200 times sweeter than table sugar (sucrose) |
Safe Levels of Sweeteners
The FDA assesses the safety of a sweetener by evaluating the available safety information about the sweetener to identify potential hazards and determine a safe level of exposure. During pre-market review, the FDA established an acceptable daily intake (ADI) level for each of the six sweeteners approved as food additives. An ADI is the amount of a substance considered safe to consume each day over the course of a person’s lifetime. The ADI is typically based on evaluating toxicological studies to determine the highest appropriate experimental exposure dose level in animal studies that was shown to cause no adverse effect, multiplied by an appropriate safety factor. In general, a safety factor is intended to provide an adequate margin of safety for consumers by accounting for variability, such as differences between animals and humans and differences in sensitivity among humans.
For each of these sweeteners, the FDA determined that the estimated daily intake of the substance would not exceed the ADI, even when considering high exposure estimates. An additive does not pose safety concerns if the estimated daily intake is less than the ADI. In the case of steviol glycosides, an ADI was established by the Joint Food and Agricultural Organization/World Health Organization Expert Committee on Food Additives (JECFA). An ADI has not been specified for monk fruit or thaumatin. An ADI may not be necessary for several reasons, including evidence of the ingredient's safety at levels well above the amounts needed to achieve the desired effect (such as sweetening) in food.
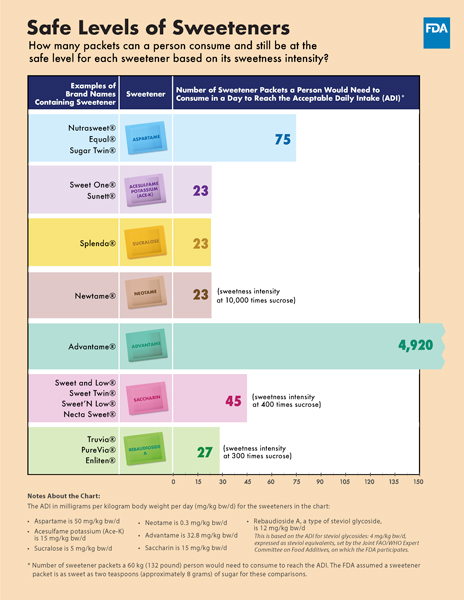
The chart below shows the safe limit for each sweetener and the amount a person would need to consume to equal that limit based on its sweetness intensity.
How many packets can a person consume and still be at the safe level for each sweetener based on its sweetness intensity?
| Examples of Brand Names Containing Sweetener | Sweetener | Number of Sweetener Packets a Person Would Need to Consume in a Day to Reach the Acceptable Daily Intake (ADI)* |
|---|---|---|
| Nutrasweet®, Equal®, Sugar Twin® | Aspartame | 75 |
| Sweet One®, Sunett® | Acesulfame Potassium (Ace-K) | 23 |
| Splenda® | Sucralose | 23 |
| Newtame® | Neotame | 23 (sweetness intensity at 10,000 times sucrose) |
| Advantame® | Advantame | 4,920 |
| Sweet and Low®, Sweet Twin®, Sweet'N Low®, Necta Sweet® | Saccharin | 45 (sweetness intensity at 400 times sucrose) |
| Truvia®, PureVia®, Enliten® | Rebaudioside A | 27 (sweetness intensity at 300 times sucrose) |
Notes About the Chart:
The ADI in milligrams per kilogram body weight per day (mg/kg bw/d) for the sweeteners in the chart:
- Aspartame is 50 mg/kg bw/d
- Acesulfame potassium (Ace-K) is 15 mg/kg bw/d
- Sucralose is 5 mg/kg bw/d
- Neotame is 0.3 mg/kg bw/d
- Advantame is 32.8 mg/kg bw/d
- Saccharin is 15 mg/kg bw/d
- Rebaudioside A, a type of steviol glycoside, is 12 mg/kg bw/d. This is based on the ADI for steviol glycosides: 4 mg/kg bw/d, expressed as steviol equivalents, set by the Joint FAO/WHO Expert Committee on Food Additives, on which the FDA participates.
* Number of sweetener packets a 60 kg (132 pound) person would need to consume to reach the ADI. The FDA assumed a sweetener packet is as sweet as two teaspoons (approximately 8 grams) of sugar for these comparisons.
Other Types of Sweeteners Allowed in the U.S.
The FDA permits the use of sugar alcohols, another class of sweeteners, as sugar substitutes. Examples include sorbitol, xylitol, lactitol, mannitol, erythritol, and maltitol. The sweetness of sugar alcohols varies from 25% to 100% as sweet as sugar. Sugar alcohols are slightly lower in calories than sugar and do not promote tooth decay or cause a sudden increase in blood glucose. They are used to sweeten foods including sugar-free candies, cookies, and chewing gums.
In 2023, the FDA reviewed a scientific paper about possible cardiovascular effects related to consuming erythritol and determined that the observational studies cited in the paper did not establish a causal link between consuming erythritol and the observed effects. The FDA will continue to monitor and review new information on erythritol, and other sweeteners, as it becomes available.
Another class of sweeteners are sugars that are metabolized differently than traditional sugars. These sugars meet the chemical definition of a sugar, but they are metabolized, or used by the body, differently than traditional sugars like sucrose. The FDA has evaluated GRAS notices for D-allulose (also referred to as D-psicose), D-tagatose, and isomaltulose and has no questions about the GRAS conclusions under certain intended conditions of use. The FDA’s response letters on D-allulose (also referred to as D-psicose), D-tagatose, and isomaltulose are available at the agency’s GRAS Notice Inventory website.
Sweeteners that are Not Allowed in the U.S.
The FDA prohibits the use of cyclamates and their salts (such as calcium cyclamate, sodium cyclamate, magnesium cyclamate, and potassium cyclamate) in the U.S. Whole-leaf and crude stevia extracts are subject to an Import Alert. They are also not permitted for use as sweeteners. These forms of stevia differ from certain highly purified steviol glycosides obtained from stevia leaves, which have been the subjects of GRAS notices; the FDA has not objected to the use as sweeteners of these highly refined substances.