FY21/22 Sample Collection and Analysis of Imported Honey for Economically Motivated Adulteration
<< Economically Motivated Adulteration
The U.S. Food and Drug Administration (FDA) conducted an assignment to collect and test imported honey in 2021 and 2022 for economically motivated adulteration. The purpose of the assignment was to identify products that contained undeclared added sweeteners and prevent them from being distributed in the U.S. market.
The FDA recognizes that the term “honey” commonly refers to the thick, sweet, syrupy substance that bees make from the nectar of plants or their secretions and store in honeycombs (see FDA’s Proper Labeling of Honey and Honey Products: Guidance for Industry).
When companies add undeclared cheaper sweeteners to honey, such as syrups derived from cane, corn, rice or sugar beets – and label the product as “honey” – they do so to lower their production costs, but consumers still pay the full price for what is deceptively labeled as honey, with the additional profit going to the companies. This is fraud, or “economically motivated adulteration,” the regulatory term used by the FDA. Simply put, consumers are being cheated.
Studies suggest that economically motivated adulteration of honey is somewhat prevalent, with findings of 13% in Canada and 14% in the European Union, respectively, in recent years.[1],[2] More than 70% of the honey consumed in the U.S. is imported.[3] Given these data, the FDA conducted this assignment to better understand the extent of economically motivated adulteration among imported honey and to identify violative products and prevent them from being distributed in the U.S. market.
Methodology
From January 2021 to March 2022, the agency collected and tested 144 samples of imported honey to determine whether they contained undeclared added sweeteners. Each sample was made up of three subsamples, with each subsample containing at least 240 milliliters of honey (about a cup). FDA field personnel collected all the samples from either bulk or retail packaged shipments labeled as “honey.” Retail packaged samples consisted of individual jars or other containers intended for retail. Bulk shipment samples were collected typically from barrels or drums, with the use of sterile equipment and aseptic procedures.
The honey evaluated for this assignment was imported from 32 countries. About 40% of the samples originated in India and Vietnam, the top two sources of honey imported by the U.S. in four of the last five years (i.e., from 2017 to 2021).[4] The FDA collected all the samples in import status, which refers to products collected at ports of entry or other locations where they are held prior to being released into domestic commerce.
The FDA used Stable Carbon Isotope Ratio Analysis (SCIRA) to analyze the organic composition of each honey sample to determine whether the honey was in compliance. When the agency found a subsample to have carbon isotope values atypical to those of authentic honey, the agency classified the associated sample as violative/noncompliant.
Findings
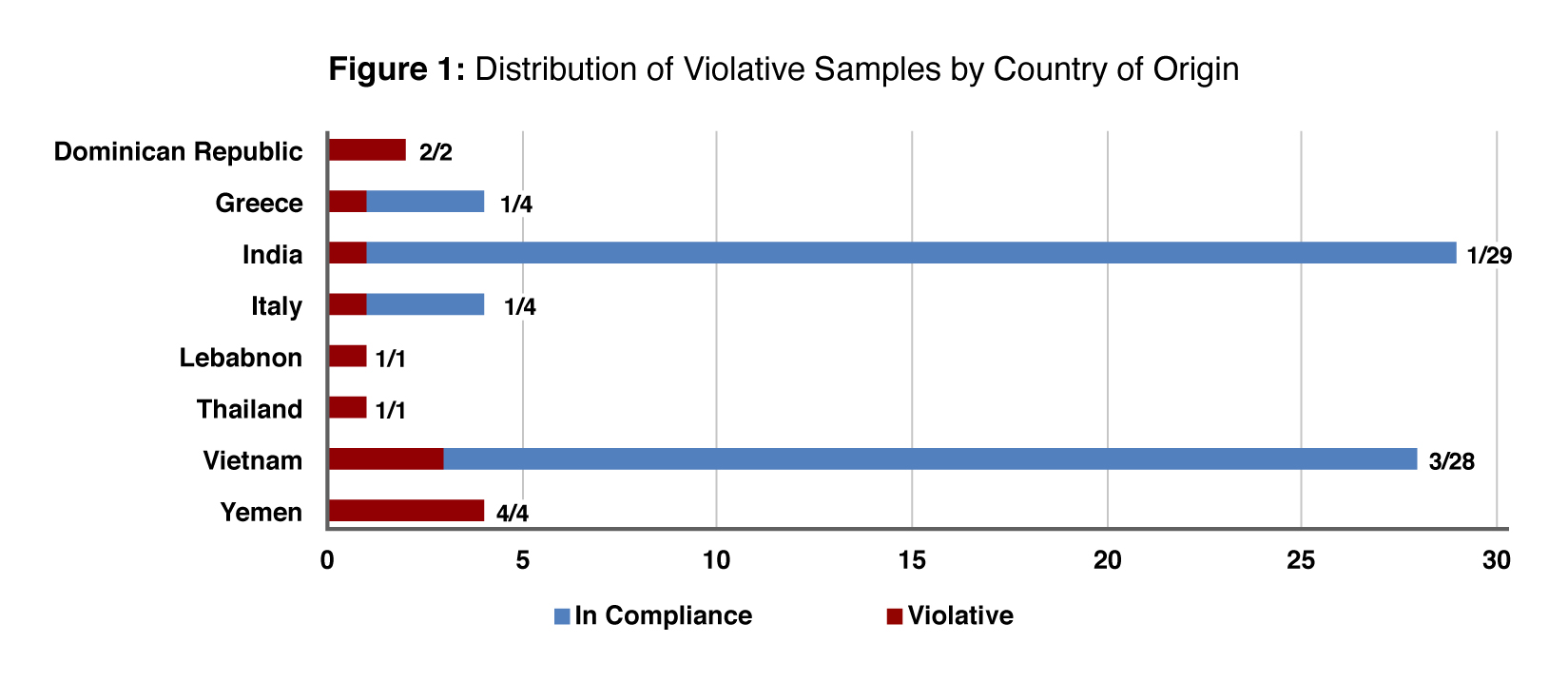
Of the 144 import samples collected and tested, the FDA found 14 (10%) to be violative. The remainder were in compliance. View the full, published dataset.
A breakdown of the findings by country of origin is provided in the chart below (Figure 1). The agency cautions against making inferences or comparisons based solely on the Figure 1 data in view of the small sample sizes and the fact that the agency did not design the assignment for such purposes.
Of the 144 import samples, 75 were collected from bulk shipments and 69 were collected from retail shipments. The difference in the adulteration rate of the bulk shipment samples versus the retail shipment samples was undetectable based on the available data (P-value = 0.58).[5]
Follow-Up Actions
When the FDA found a sample to be violative, the agency refused entry of the shipment into the U.S. and placed the associated company and product on Import Alert (IA) 36-01. Products listed on the import alert are subject to Detention without Physical Examination. For those products to be admitted into the U.S., the company is required to provide evidence to the FDA to overcome the appearance of a violation, such as the test results of a third-party laboratory, verifying that the product does not contain added sweeteners. The agency placed violative firms on IA 36-01 during this assignment.
Discussion
The results of this assignment reaffirm the need for the agency to continue to test imported honey for economically motivated adulteration under the agency’s risk-based import entry screening program. Violative samples are subject to compliance actions, such as recalls and import refusal, consistent with the FDA’s mission to ensure that food is safe, wholesome, and properly labeled. When appropriate, the agency may consider pursuing criminal investigations. The FDA also collaborates with international counterparts to detect and combat economically motivated adulteration related to imported products, including imported honey.
Companies that import honey for retail sale, or that use honey as an ingredient in their food manufacturing, can help protect consumers by ensuring the authenticity of the honey. When other sweeteners are added to honey, the Federal Food, Drug & Cosmetic (FD&C) Act requires that the resulting product be labeled as a “blend,” and not as “honey,” and have an ingredient statement. Labeling on packaged food in interstate commerce is required to be truthful and not misleading, according to the FD&C Act.
For additional information on the FDA’s work to combat food fraud, please visit the agency’s Economically Motivated Adulteration (Food Fraud) page.
[1] Report: Honey authenticity surveillance results (2019 to 2020). Canadian Food Inspection Agency.
[2] Honey (2015-17). Technical Report by the Joint Research Centre, European Commission.
[3] U.S. Honey Production and Trade. Table No. 46. April 15, 2022. USDA Economic Research Service: Sugar and Sweeteners Yearbook Tables.
[4] Ibid, Table No. 48.
[5] Fisher’s exact test