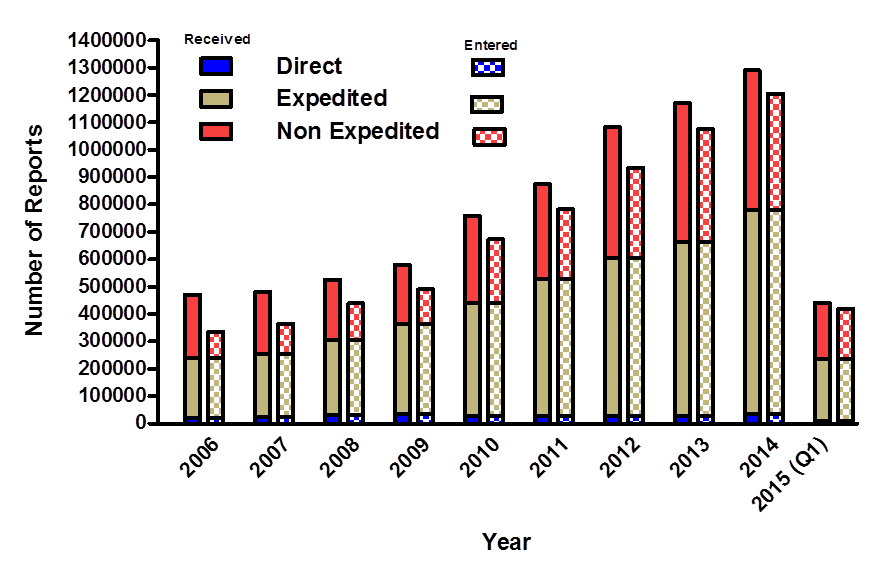
Reports Received and Reports Entered into FAERS by Year
(As of November 2015)
Back to FAERS Statistics Main Page
These data describe total numbers of reports we received for drugs and therapeutic biologic products and the number of reports we entered into the FAERS database. Not all of the reports that FDA receives for drug and therapeutic biologic products are entered into the FAERS database. At the present time, we are entering reports of the following types:
- Reports submitted directly to FDA (not submitted through manufacturers)
- Reports submitted on 3500A (or CIOMS) paper forms by manufacturers that are categorized as:
- expedited reports
- non-expedited reports of serious adverse events, or
- non-expedited reports of non-serious adverse events, for new molecular entity (NME) products within the first 3 years following FDA approval
- Reports submitted electronically by manufacturers regardless of category.
A manufacturer's expedited report is a report that contains at least one adverse event that is not currently described in the product labeling and for which the patient outcome is serious. A manufacturer's non-expedited report is a report that did not meet the criteria for an expedited report. Manufacturers submit non-expedited reports to FDA quarterly for the first three years after approval, and annually thereafter.
Figure 1. This figure illustrates the number of reports received (solid bars) and entered (checkered bars) into AERS by type of report since the year 2006 through the first quarter of 2015.
| FAERS YEAR | Expedited | DIRECT | Non Expedited Entered | Total Entered | Non Expedited Received | Total Received |
|---|---|---|---|---|---|---|
| 2006 | 219,217 | 20,979 | 95,555 | 335,751 | 230,065 | 470,261 |
| 2007 | 229,982 | 23,032 | 110,405 | 363,419 | 228,206 | 481,220 |
| 2008 | 274,281 | 32,896 | 132,690 | 439,867 | 218,205 | 525,382 |
| 2009 | 330,383 | 34,165 | 126,177 | 490,725 | 216,264 | 580,812 |
| 2010 | 409,547 | 28,946 | 234,646 | 673,169 | 320,341 | 758,834 |
| 2011 | 499,155 | 28,045 | 255,275 | 782,475 | 346,745 | 873,945 |
| 2012 | 577,852 | 29,026 | 326,637 | 933,515 | 475,993 | 1,082,871 |
| 2013 | 635,206 | 28,386 | 411,622 | 1,075,214 | 506,512 |
1.170,104
|
| 2014 | 746,584 | 34,246 | 423,855 | 1,204,685 | 508,303 | 1,289,133 |
| 2015(Q1) | 227,347 | 9,824 | 181,856 | 419,027 | 203,442 | 440,613 |