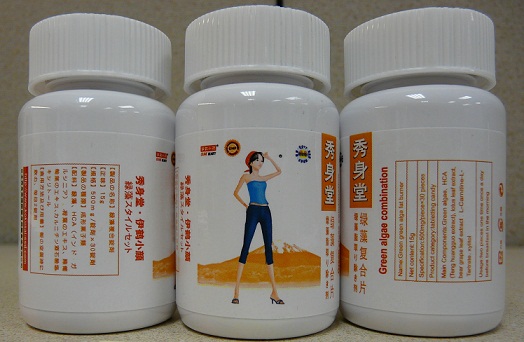
Public Notification: Green Algae Combination by Crane Beauty contains hidden drug ingredient
[3-26-2015] The Food and Drug Administration (FDA) is advising consumers not to purchase or use Green Algae Combination by Crane Beauty, a product promoted for weight loss. This product was identified by FDA during an examination of international mail shipments.
FDA laboratory analysis confirmed that Green Algae Combination by Crane Beauty contains lorcaserin, a controlled substance and the active ingredient in the FDA-approved prescription drug Belviq®, used for chronic weight management in some overweight or obese adults. Lorcaserin may cause psychiatric disturbances and impairments in attention or memory. Also, concomitant use of lorcaserin with certain medications, such as, but not limited to, drugs commonly used to treat depression and migraine, may result in a potentially life-threatening drug interaction.
Health care professionals and patients are encouraged to report adverse events or side effects related to the use of this product to FDA's MedWatch Safety Information and Adverse Event Reporting Program:
- Complete and submit the report online at MedWatch Online Voluntary Reporting Form, or;
- Download and complete the form, then submit it via fax at 1-800-FDA-0178.
Note: This notification is to inform the public of a growing trend of dietary supplements or conventional foods with hidden drugs and chemicals. These products are typically promoted for sexual enhancement, weight loss, and bodybuilding and are often represented as being “all natural.” FDA is unable to test and identify all products marketed as dietary supplements that have potentially harmful hidden ingredients. Consumers should exercise caution before purchasing any product in the above categories.
For more information: