What's New in Drugs@FDA
March 15, 2012
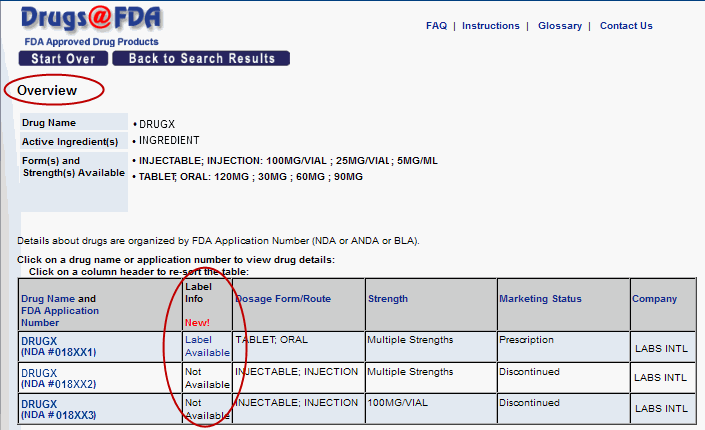
- New "Label Info" column on Overview page. [Figure 1]
A new column tells you one screen earlier if a labels are available for applications with identical drug names. You can click on "Label Available" to go directly to the Label and Approval History page.
- Easier Month and Year selection in "Drug Approval Reports by Month."
You can now use a dropdown menu to select the month or year instead of typing.
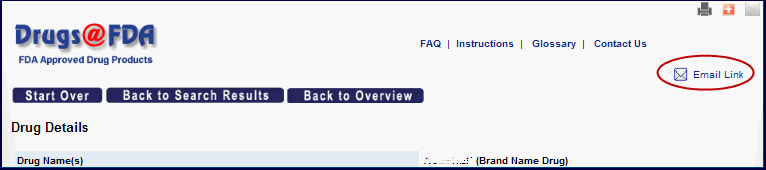
- Want to mail someone a link to your search results? Use the new "Email link" icon. [Figure 2]
Clicking the icon opens a new message in your default email client. It supplies the subject "A Link from Drugs@FDA" and supplies a URL for you to copy or send. When you later click on the URL, you will get the current information for that search, which may have changed in the meantime.
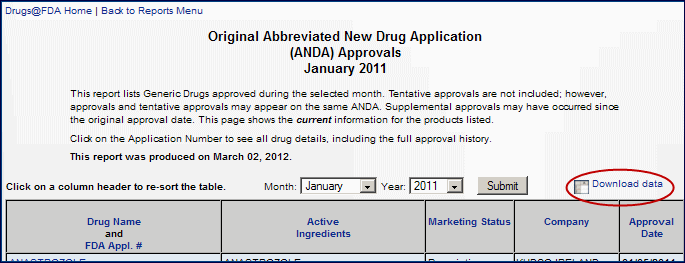
- Want to download results to a spreadsheet? Use the new "Download data" icon. [Figure 3]
By clicking the "Download data" icon you can save or open a table as a CSV (comma separated values) format for Excel or other applications. This feature is available on the following pages:- Label and Approval History (You can download the Approval History.)
- Therapeutic Equivalents
- Other OTC Drugs with the Same Active Ingredient, Strength and Dosage Form/Route
- Drug Approval Reports by Month
Figure 1. New "Label Info" column on Overview page


 >
>