Patient-Focused Drug Development

The Oncology Center of Excellence (OCE) Patient-Focused Drug Development (PFDD) program fosters collaboration between FDA Centers and external stakeholders involved in patient outcomes research in cancer populations.
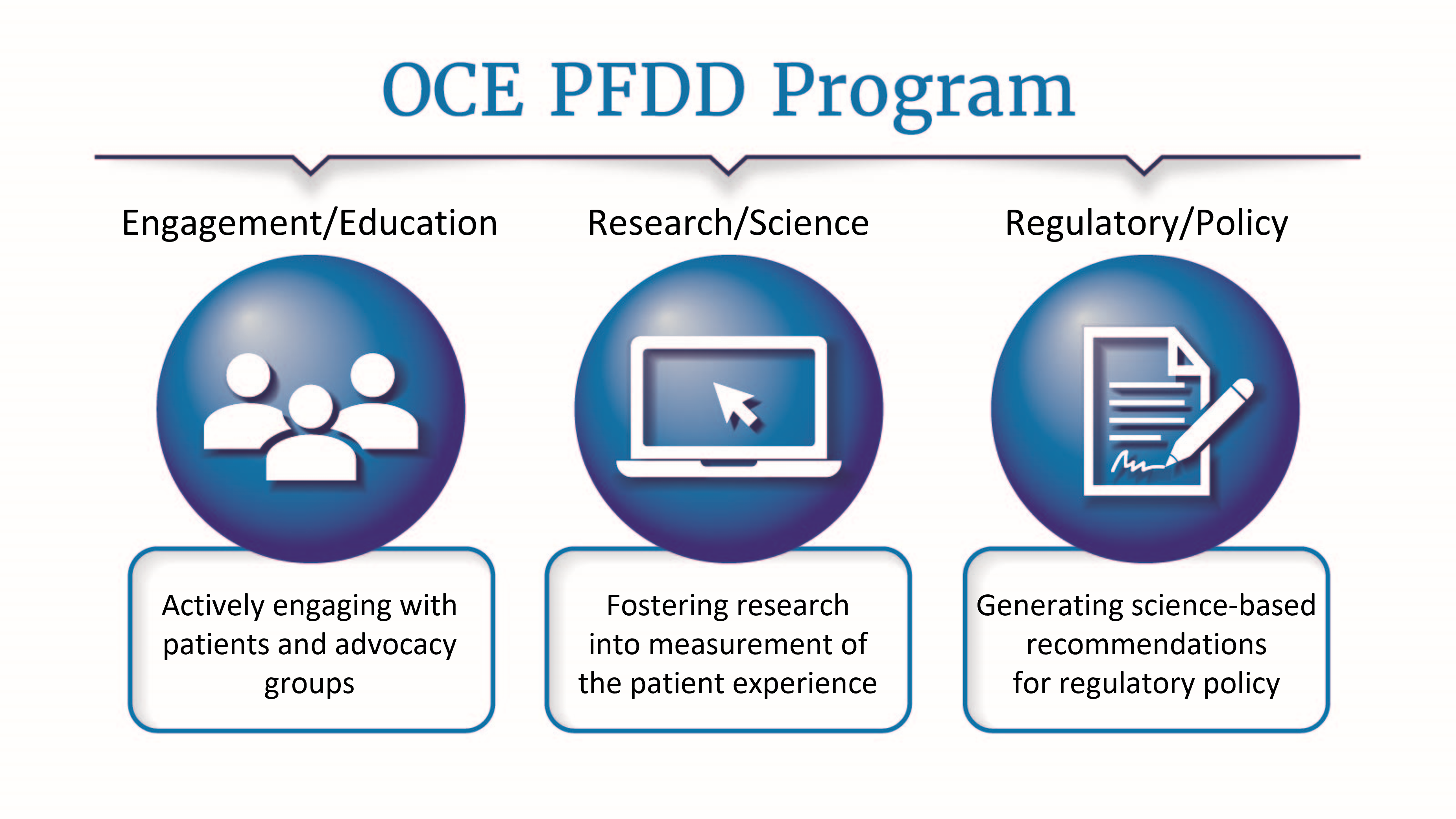
Cancer patients experience disease symptoms and symptomatic treatment side effects that can impact their ability to function and other aspects of their health-related quality of life. The PFDD Program identifies rigorous methods to assess the patient experience which complements existing survival and tumor information to provide additional evidence about the effects of cancer therapies on patients.
The program focuses on three key areas:
In 2021, the OCE along with CDER and CBER published a guidance for industry providing recommendations for the collection of a core set of patient-reported clinical outcomes in cancer clinical trials and related considerations for instrument selection and trial design: “Core Patient-Reported Outcomes in Cancer Clinical Trials.”
PFDD Team
PFDD Lead: Vishal Bhatnagar, MD
PFDD Regulatory Program Manager: Esther Park, PharmD
Contact Information
Contact: OCEPFDD@fda.hhs.gov
Projects
- Collaborator: Kaiser Permanente, Division of Research – PANcreatic cancer Patient Reported Outcomes using the Electronic medical record (PanPROE)
- Collaborator: Northwestern University and University of Tasmania – Evaluation of a global item for side effect bother
- Collaborator: Duke University - Evaluating physical functioning using patient-reported outcome measures: How does the question form and recall period influence patients’ interpretation?
- Collaborator: Mayo Clinic and Yale University - Quantifying physical function in cancer patients undergoing chemotherapy using clinician- and patient-reports along with wearable device data
- Collaborator: Mayo Clinic and Yale University - Longitudinal analysis & visualization of patient-reported physical function & symptom data
- Collaborator: Mayo Clinic and Yale University - Qualitative techniques to define meaningful within-patient change in symptoms of advanced cancer patients
- Collaborator: National Cancer Institute– Advancing the use of digital health technologies and digital symptom reporting for patient-reported outcome measures
Working Groups
- National Cancer Institute (NCI) Cancer Moonshot Tolerability Consortium Steering Committee
- Patient-Reported Outcomes Tools: Engaging Users & Stakeholders (PROTEUS) consortium
- Setting International Standards in Analyzing Patient-Reported Outcomes and Quality of Life Endpoints Data (SISAQOL)
- Standard Protocol Items: Recommendations for Interventional Trials Patient-Reported Outcomes (SPIRIT-PRO)
Upcoming Events:
Past Events:
Clinical Outcomes Assessment in Clinical Cancer Trials (COA-CCT) Workshops
October 8, 2025: 10th COA-CCT Workshop
June 25, 2024: 9th COA-CCT Workshop
June 27, 2023: 8th COA-CCT Workshop
June 29, 2022: 7th COA-CCT Workshop
July 21 and 23, 2021: 6th COA-CCT Workshop
July 17, 2020: 5th COA-CCT Workshop with American Society of Clinical Oncology
July 12, 2019: 4th COA-CCT Workshop with American Society of Clinical Oncology
June 22, 2018: 3rd COA-CCT Workshop with American Society for Clinical Oncology
April 25, 2017: 2nd COA-CCT Workshop with the Critical Path Institute
April 26, 2016: COA-CCT Workshop with the Critical Path Institute
Other Events
May 11-12, 2021: Meeting of the Pediatric Oncology Subcommittee of the Oncologic Drugs Advisory Committee
October 8, 2019: 3rd Partners in Progress
November 27, 2018: 2nd Partners in Progress
November 13, 2017: 1st Partners in Progress
The OCE PFDD Program continuously publishes on topics to advance patient-focused drug development. Recent articles are listed below:
Murugappan MN, King-Kallimanis BL, Bhatnagar V, Kanapuru B, Farley JF, Seifert RD, et al. Measuring Frailty Using Patient-Reported Outcomes (PRO) Data: A Feasibility Study in Patients with Multiple Myeloma. Quality of Life Research. 2023 Mar 20:1-2.
Thanarajasingam G, Kluetz PG, Bhatnagar V, Brown A, Cathcart-Rake E, Diamond M, et al. Integrating 4 Measures to Evaluate Physical Function in Patients with Cancer (In4M): Protocol for a prospective study. medRxiv. 2023 Mar 9.
Piccinin C, Basch E, Bhatnagar V, Calvert M, Campbell A, Cella D, et al. Recommendations on the use of item libraries for patient-reported outcome measurement in oncology trials: findings from an international, multidisciplinary working group. The Lancet Oncology. 2023 Feb 1;24(2):e86-95.
Bhatnagar V, Dutcus C, Ghiorghiu S, Kluetz P, Jones L, McJunkins KR, et al. Supporting a Patient-Centric Approach to Dose Optimization in Oncology: The Essential Role of Patient-Reported Outcomes (PROs). Friends of Cancer Research White Paper. 2022.
Bhatnagar V, Kluetz PG. Encouraging Rigorous Patient-Generated Data All Along the Drug Development Continuum. Journal of the National Cancer Institute. 2022 Oct;114(10):1313-4.
King-Kallimanis BL, Bhatnagar V, Horodniceanu EG, Chen TY, Kluetz PG. Timing is everything: The importance of patient-reported outcome assessment frequency when characterizing symptomatic adverse events. Clinical Trials. 2022 Jun;19(3):267-73.
Fiero MH, Roydhouse JK, Bhatnagar V, Chen TY, King-Kallimanis BL, Tang S, et al. Time to deterioration of symptoms or function using patient-reported outcomes in cancer trials. The Lancet Oncology. 2022 May 1;23(5):e229-34.
Chen TY, King-Kallimanis BL, Merzoug L, Horodniceanu EG, Fiero MH, Gao JJ, et al. US Food and Drug Administration Analysis of Patient-Reported Diarrhea and Its Impact on Function and Quality of Life in Patients Receiving Treatment for Breast Cancer. Value in Health. 2022 Apr 1;25(4):566-70.
Murugappan MN, King‐Kallimanis BL, Mangir C, Howie L, Bhatnagar V, Beaver JA, et al. Floor and ceiling effects in the EORTC QLQ‐C30 Physical Functioning subscale among patients with advanced or metastatic breast cancer. Cancer. 2022 Feb 15;128(4):808-18.
Murugappan MN, King-Kallimanis BL, Reaman GH, Bhatnagar V, Horodniceanu EG, Bouchkouj N, et al. Patient-reported outcomes in pediatric cancer registration trials: A US Food and Drug Administration perspective. JNCI: Journal of the National Cancer Institute. 2022 Jan;114(1):12-9.
King-Kallimanis BL, Lederer NM, Kim J, Nair A, Horodniceanu E, Bhatnagar V, et al. Patient-reported outcomes after treatment discontinuation: commercial clinical trial data from four cancer types. Value in Health. 2021 Sep 1;24(9):1302-7.
Fernandes LL, Zhou J, Kanapuru B, Horodniceanu E, Gwise T, Kluetz PG, et al. Review of patient-reported outcomes in multiple myeloma registrational trials: highlighting areas for improvement. Blood Cancer Journal. 2021 Aug 31;11(8):148.
Calvert M, King M, Mercieca-Bebber R, Aiyegbusi O, Kyte D, Slade A, et al. SPIRIT-PRO Extension explanation and elaboration: guidelines for inclusion of patient-reported outcomes in protocols of clinical trials. BMJ open. 2021 Jun 1;11(6):e045105.
Roydhouse JK, Mishra-Kalyani PS, Bhatnagar V, Gutman R, King-Kallimanis BL, Sridhara R, et al. Does knowledge of treatment assignment affect patient report of symptoms, function, and health status? An evaluation using multiple myeloma trials. Value in Health. 2021 Jun 1;24(6):822-9.
Kluetz PG, King-Kallimanis BL, Suzman D, Chang E, Brave MM, Weinstock C, et al. Advancing assessment, analysis, and reporting of safety and tolerability in cancer trials. JNCI: Journal of the National Cancer Institute. 2021 May;113(5):507-8.
Kluetz PG, Bhatnagar V. The FDA's patient-focused drug development initiative. Clinical Advances in Hematology & Oncology: H&O. 2021 Feb 1;19(2):70-2.
Bhatnagar V, Hudgens S, Piault‐Louis E, Jones L, Beaver JA, Lyerly HK, et al. Patient‐reported outcomes in oncology clinical trials: Stakeholder perspectives from the accelerating anticancer agent development and validation workshop 2019. The Oncologist. 2020 Oct;25(10):819-21.
Roydhouse JK, King-Kallimanis BL, Roy P, Weinstock C, Krol D, Daniels SR, et al. Exploration of baseline patient-reported side effect bother from cancer therapy. Clinical Trials. 2020 Jun;17(3):332-7.
Roydhouse JK, Gutman R, Bhatnagar V, Kluetz PG, Sridhara R, Mishra‐Kalyani PS. Analyzing patient‐reported outcome data when completion differs between arms in open‐label trials: an application of principal stratification. Pharmacoepidemiology and Drug Safety. 2019 Oct;28(10):1386-94.
King-Kallimanis BL, Howie LJ, Roydhouse JK, Singh H, Theoret MR, Blumenthal GM, et al. Patient reported outcomes in anti-PD-1/PD-L1 inhibitor immunotherapy registration trials: FDA analysis of data submitted and future directions. Clinical Trials. 2019 Jun;16(3):322-6.
Roydhouse JK, King-Kallimanis BL, Howie LJ, Singh H, Kluetz PG. Blinding and patient-reported outcome completion rates in US food and drug administration cancer trial submissions, 2007–2017. JNCI: Journal of the National Cancer Institute. 2019 May 1;111(5):459-64.
Roydhouse JK, Fiero MH, Kluetz PG. Investigating potential bias in patient-reported outcomes in open-label cancer trials. JAMA oncology. 2019 Apr 1;5(4):457-8.
Kluetz PG, Kanapuru B, Lemery S, Johnson LL, Fiero MH, Arscott K, et al. Informing the tolerability of cancer treatments using patient-reported outcome measures: summary of an FDA and critical path institute workshop. Value in Health. 2018 Jun 1;21(6):742-7.
Kluetz PG, O'Connor DJ, Soltys K. Incorporating the patient experience into regulatory decision making in the USA, Europe, and Canada. The Lancet Oncology. 2018 May 1;19(5):e267-74.
Kim J, Singh H, Ayalew K, Borror K, Campbell M, Johnson LL, et al. Use of PRO Measures to Inform Tolerability in Oncology Trials: Implications for Clinical Review, IND Safety Reporting, and Clinical Site Inspections. Clinical Cancer Research. 2018 Apr 15;24(8):1780-4.
Kluetz PG, Chingos DT, Basch EM, Mitchell SA. Patient-reported outcomes in cancer clinical trials: measuring symptomatic adverse events with the National Cancer Institute’s Patient-Reported Outcomes Version of the Common Terminology Criteria for Adverse Events (PRO-CTCAE). American Society of Clinical Oncology Educational Book. 2016 May;36:67-73.
Kluetz PG, Slagle A, Papadopoulos EJ, Johnson LL, Donoghue M, Kwitkowski VE, et al. Focusing on core patient-reported outcomes in cancer clinical trials: symptomatic adverse events, physical function, and disease-related symptoms. Clinical Cancer Research. 2016 Apr 1;22(7):1553-8.
This program is part of the OCE Scientific Collaborative. Click here for more information.