Jobs in Oncology/Hematology
Transformative careers in FDA Oncology
FDA is recruiting oncologists and hematologists to serve in the dynamic, highly challenging, and innovative atmosphere of oncology drug development and research.
Who We Are
Meet Dr. Nicole Gormley, Division Director of Hematologic Malignancies 2, as she
discusses what’s satisfying about the work she's doing in the multiple myeloma field at FDA Oncology.
The Office of Oncologic Diseases (OOD) consists of oncologists from all career backgrounds, stages, and fields of expertise, including medical, surgical, pediatric, radiation, and hematologic oncologists. There are five divisions of therapeutic areas and one nonclinical division within OOD.
We may spend up to eight hours weekly seeing patients in clinic to maintain our clinical skills. Area oncology centers include Johns Hopkins, Sibley Memorial Hospital, University of Maryland, George Washington University, Georgetown University, National Cancer Institute (NIH), Veterans Administration (VA), and Walter Reed National Military Medical Center.
FDA oncologists trained and worked at various academic centers throughout the country. A few of the institutions include:
- Children’s Hospital of Philadelphia
- Johns Hopkins Sidney Kimmel Cancer Center
- MD Anderson Cancer Center
- Memorial Sloan Kettering Cancer Center
- National Cancer Institute
- University of Chicago
- Vanderbilt University Medical Center
- and many more
What We Do
As oncologists and hematologists at the FDA, we engage researchers, patients, global regulators, and educators in a multifaceted approach to improve cancer care. Our commitment to public health, collaboration, and patient-centered regulatory decision-making drives our passion for advancing the availability of safe and effective drugs and biologics to patients with cancer. This manifests in the following six areas of activity.
Regulatory Review
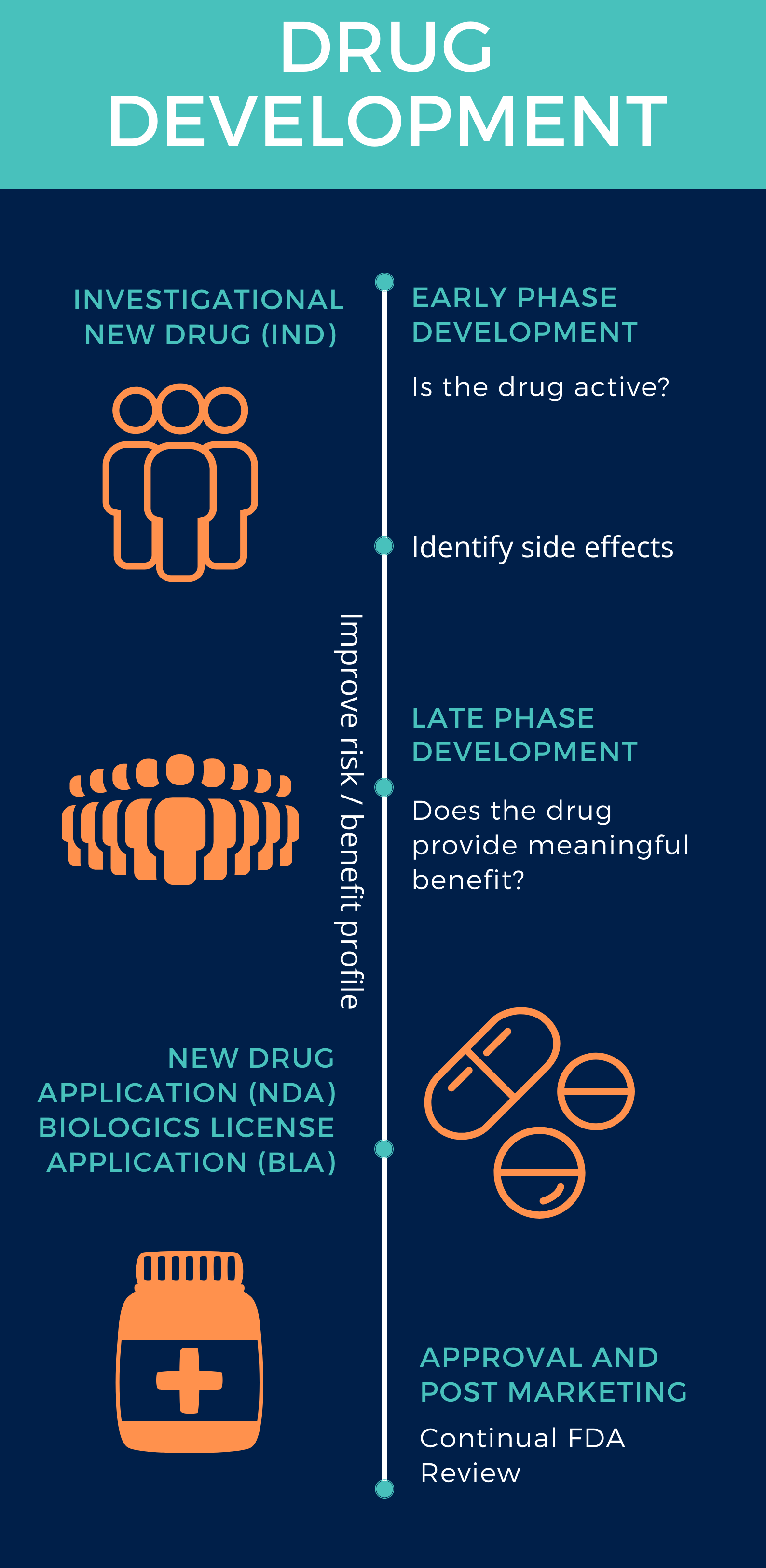
Learn more about Investigational New Drugs (INDs) and Project Facilitate, a pilot program to assist oncology healthcare providers or regulatory professionals in requesting access to investigational therapies for patients with cancer.
Still curious? Read the Top 10 Myths About FDA’s Office of Hematology and Oncology Products.
Research
We are very active in research. An analysis of recent oncology scientific publications revealed that our papers are enriched for high-impact publications, and have nearly two times the number of citations as an average NIH-funded paper. FDA oncology staff has contributed to numerous publications in scientific literature which can be found in PubMed including The New England Journal of Medicine, Lancet Oncology, Nature Reviews Clinical Oncology, Journal of Clinical Oncology, JAMA Oncology, and more. Visit OCE Publications to browse our articles.
Education
Through open communication, education, and outreach, we aim to provide transparency and insight into the FDA’s role in oncology drug development, and the multidisciplinary and team-based approach the FDA takes in this process.
Read more in the ASCO Post article and learn about Project Socrates, one of the many Oncology Center of Excellence (OCE) initiatives.
External Engagement
Engagement with international regulatory agencies: As a result of cancer drug development being global, Project Orbis provides international access to safe and effective drugs earlier through concurrent submission and review of oncology products.
Engagement with patients, caregivers, and their healthcare providers: Project Patient Voice is patient-reported symptom data collected from clinical trials. The data will be available in an online interactive platform to help patients, and their healthcare providers, understand the patient experience, risks, and benefits of a drug.
Regulatory Policy
FDA Oncology actively develops Guidances for Industry* on topics such as patient-reported outcomes in cancer clinical trials, optimizing the dosage of drugs and biological products for oncological diseases, cancer clinical trial eligibility criteria, regulatory considerations for use of minimal residual disease in hematologic malignancies, and clinical trial endpoints for the approval of cancer drugs and biologics.
*Guidance documents represent FDA's current thinking on a topic, describing FDA's interpretation of our policy on a regulatory issue.
Professional Development
At the FDA, we can further develop our career with pursuits in research or clinical care. Some of us continue to see patients for up to 8 hours a week in a variety of clinical settings such as precepting fellows, second-opinion clinics, continuity clinics, and inpatient medicine in academic, community, or government facilities. We also engage in many research activities, and we publish original research, review articles and commentaries, approval summaries for drugs we approve for marketing, and expert working group reports.
Join Us
Whether you are just starting out of training, or ready for a career change, we would love to connect with you!
- Contact FutureofHemeOnc@fda.hhs.gov if interested in job opportunities or to learn about benefits such as loan repayment and 1:1 matching for government retirement contributions.
- Learn about the Quality of Worklife at FDA, including benefits and flexible telework options.
- And join our community on the X platform @FDAOncology!