Reverse Osmosis
[Previous Chapter] [Table of Contents] [Next Chapter]
WELFARE PUBLIC HEALTH SERVICE
FOOD AND DRUG ADMINISTRATION
*ORA/ORO/DEIO/IB*
Related Program Areas:
Drugs, Medical Devices and Diagnostic
Products
ITG SUBJECT: REVERSE OSMOSIS
Introduction
Reverse osmosis (RO) has been known for more than a century, but it did not become a commercial process until the early sixties when a special membrane was developed (1,2,3,4). Because RO operates at a comparatively low temperature and is relatively energy efficient, it is employed in various applications, e.g., desalination, treatment of waste water, reclamation of minerals, concentration of whey and other food products, and purification of water (5,6). In recent years, RO has been used increasingly in making processed water for dialysis in hospitals and for certain cosmetics and drugs by pharmaceutical manufacturers (7,8). In addition to these applications, RO is capable of producing water of sufficient purity to be used as Water For Injection (WFI) and for the preparation of parenteral solutions (9,10,11,12). This ITG will focus on the chemical and microbiological quality of water produced by reverse osmosis.
Definition and Operating Principle
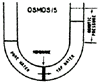
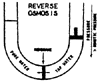
Reverse osmosis is a process which uses a membrane under pressure to separate relatively pure water (or other solvent) from a less pure solution. When two aqueous solutions of different concentrations are separated by a semi-permeable membrane, water passes through the membrane in the direction of the more concentrated solution as a result of osmotic pressure (Figure 1). If enough counter pressure is applied to the concentrated solution to overcome the osmotic pressure, the flow of water will be reversed (Figure 2).
Water molecules can form hydrogen bonds in the RO membrane and fit into the membrane matrix. The water molecules that enter the membrane by hydrogen bonding can be pushed through under pressure. Most organic substances with a molecular weight over 100 are sieved out, i.e., oils, pyrogens and particulates including bacteria and viruses (13).
Salt ions, on the other hand, are rejected by a mechanism related to the valence of the ion. Ions are repelled by dielectric interactions; ions with higher charges are repelled to a greater distance from the membrane surface. Monovalent ions such as chloride ions will not be rejected as efficiently as, for example, divalent sulfate ions. The nominal rejection ratio of common ionic salts is 85 - 98%.
Membrane
The majority of the commercially manufactured RO membranes are made from cellulose acetate, polysulfonate, and polyamide. Many other kinds of membrane made of a single polymer or a copolymer are also available for specific purposes. The membrane consists of a skin about 0.25 microns and a support layer about 100 microns. The skin is the active barrier and primarily allows water to pass through.
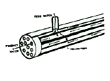
Two types of RO construction are commonly used: 1. spiral wound ---sheets of membrane sandwiched with mesh spacers are connected and wound around a permeate tube; and 2. hollow fiber. Either of these modules is assembled into a pressure housing. Schematics of these two types of membrane modules (permeators) are shown below (Figures 3 and 4).
COMMON TYPES OF RO MODULES (PERMEATORS)
Operation
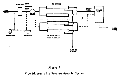
A typical reverse osmosis system is shown in the flow diagram (Figure 5) (image size 11KB). Feed water is passed through a prefilter after pH adjustment, and is pumped to the membrane modules at the designed pressure. The product water is then pumped to a storage tank and the concentrate is drained (6). Most RO units with a cellulose acetate membrane are designed to operate between 55 F - 86 F. (13 C - 30 C). In practice, the feed water may go through any one or a combination of several of the following pretreatments: sand bed, chlorinator and retention tank, anthracite filter, activated charcoal filter, degasifier, microfilter, neutralizer, and deionizer, depending on the condition of the feed water and the desired quality of the product water. Since water conditions may vary from time to time, adequate pretreatment must be provided so that the dissolved solids and the bacterial level of the feed water, after prefiltration, can be controlled within the designed limits. Two RO modules in series should be considered for parenteral solutions principally for the reduction of monovalent ions and bacterial contaminants.
FIGURE 5 FLOW DIAGRAM OF A REVERSE OSMOSIS SYSTEM
A major problem in operating RO systems is concentration polarization or fouling which is the gradual build up of rejected solute on the feed side, immediately adjacent to the membrane. A flush cycle is often used to reduce build up. The spiral wound construction is less susceptible to fouling than that of the hollow fiber unit. A membrane module lasts two to three years on the average. The shut down procedure for non-working hours should assure that minimum flow and operating pressures are continued with a timed internal flush cycle.
Quality of RO Product Water
The amount of dissolved solids in water produced by reverse osmosis is approximately a constant percentage of those in the feed water. For example, when the feed water contains 300 ppm total dissolved solids (TDS), the product water may have 15 to 30 ppm (95% and 90% rejection ratio respectively). A RO system design is based on a certain range of feed water TDS, the percentage of rejection and percentage of recovery desired. For a given system, the higher the percentage of recovery or the lower the percentage of rejection, the poorer the quality of product water becomes. A RO water purification system with a deionizer and/or several modules connected in series can produce water containing less than 0.1 ppm TDS (resistivity about 1 megohm-cm). In defining water for use parenterally, Remington's Pharmaceutical Sciences emphasizes that besides meeting USP pyrogen standards, there are several acceptable levels for total solids which may be used to evaluate or grade the product water for particular applications. The USP Monograph limits total solids to 10 PPM for WFI. However, to preclude stability problems from interfering with manufacturing, the parenteral drug industry commonly establishes a limit of 0.1 PPM or less of ionic contaminants (14).
It has been reported that bacteria can "grow" through membranes. The mechanism by which bacteria pass through a RO membrane is not known and no correlation exists between a dye leak test of the membrane and its bacterial retention efficiency. Researchers at the Center for Disease Control (CDC) conducted extensive investigations on the bacterial contamination of RO systems used in producing purified water for dialysis (15). They reported: 1. certain naturally occurring Gram- negative bacteria can multiply in relatively pure RO water; 2. thorough periodic disinfection of the entire RO system is essential in producing water with acceptable bacterial counts; 3. stagnant water in pipes down stream of the membrane is the major source of bacteria and endotoxin in the product water; and 4. the efficiency of a membrane in rejecting bacteria is better in continuous operation than in intermittent use.
Modular RO units with production rates ranging from a few to several hundred gallons per day are now available from several manufacturers. These units, when properly operated, can produce water which meets the chemical, microbiological and pyrogen test of WFI according to USP XX.
Technical Requirements of a RO System
Several basic technical requirements of a RO system are:
- Feed water should be adjusted to proper pH and be prefiltered. The amount of TDS and suspended materials in the feed water after prefiltration should be controlled within the designed limits.
- Feed water and product water should be monitored for microbiological quality. The system should be disinfected when microbiological quality levels are exceeded.
- All system components should be mechanically cleaned before disinfecting. Appropriate tests should be performed to assure that chemicals used in disinfection are completely removed from the system.
- The use of filters or ion-exchangers down stream of RO modules should be avoided.
- The RO system should be designed for continuous flow without traps, dead ends and pipe sections which may collect stagnant water.
- The chemical and microbial quality of water should be tested at predetermined intervals during a production cycle. In-line conductivity probes should be installed at key points for continuous monitoring of water quality.
- The equipment should be qualified and the RO system should be validated periodically, as well as operated and maintained according to manufacturer's instructions so that it can consistently produce water with acceptable quality.
References
- Gregor, H.P., and Gregor, C.D., "Synthetic Membrane Technology," Scientific American, July, 1978.
- Sourirajan, S., Reverse Osmosis, Logos Press, London; Academic Press, New York, N.Y., 1970.
- Karger, Barry L. et al. An Introduction to Separation Science. New York: Wiley, 1965.
- Londale, H. and Podall, T., Ed. Reverse Osmosis Membrane Research. New York: Plenum Press, 1972.
- Elias, S., "Membrane Processing," Food Engineering. Oct. 1979.
- Lacey, R. E., "Membrane Separation Process," Chem. Eng., Sept. 1972.
- Datta, R. et al. "Concentration of Antibiotics by Reverse Osmosis," Biotechnology and Bioengineering XIX, 1419-1429, 1977.
- Favero, M. S., Peterson, N. J., et al. "Gram-Negative Water Bacteria in Hemodialysis Systems," Health Laboratory Science, Vol. 12. No. 4. 1975.
- Klumb, G. H., "Reverse Osmosis - A Process in the Purification of Water for Parenteral Administration," Bulletin of the Parenteral Drug Association, Vol. 29. No. 5. 1975.
- Frith, C. F., Dawson, F. W. and Sampson, R. L., "Water for Injection USP XIX By Reverse Osmosis," Bulletin of the Parenteral Drug Association, Vol. 30, No. 2, 1976.
- Hoag, Selwyn B. and William F. Albern. "Reverse Osmosis: The Economical Production of Quality Water," Plumbing Engineer, May-June, 1977.
- Juberg, Donald L. "Application of Reverse Osmosis for the Generation of Water for Injection," Bulletin of the Parenteral Drug Association 31: 70-78, March-April, 1977.
- Belfort, G., Rotem, Y., and Katzenelson, E., "Virus Concentration Using Hollow Fiber Membranes," Water Research, Vol. 9, 1975.
- Osol, Arthur, Ed. Remington's Pharmaceutical Sciences, 16th Edition, Easton, Penn: Mack, 1980.
- Peterson, N. J., et al. Quarterly Reports, April-June, 1976, July-Sept. 1977. HHS, PHS, CDC, Phoenix, Lab. Div., Phoenix, Arizona.