The C. elegans Model in Toxicity Testing
New Approach Methods (NAMs) Main Page
Strong science underpins everything FDA does. The scientific discipline of toxicology is particularly essential to FDA’s mission and is crucial to the Agency’s ability to evaluate product safety and reach sound regulatory decisions.
New types of testing methods are being developed to identify harmful biological effects of chemicals and other substances, the dosages that cause those effects, and safe exposure limits of these products.
Historically, scientists have relied heavily on animal studies to determine if the components of a food or drug are safe. Although animal testing is still necessary in many situations, it is expensive and time-consuming. Traditional toxicology tests on mammals such as mice and rabbits can take over a year to complete. You need to house and feed the animals for a long period of time. You need veterinarians on staff to assist the regular lab employees. All this costs money, and the regulatory review process takes much longer.
To combat these limitations and the budgetary constraints associated with live animal testing, Piper Hunt, a research scientist in the Division of Toxicology in the Office of Applied Research and Safety at FDA, has started to test new technologies and alternative toxicology methods that may be more efficient and affordable. Rapidly evolving scientific advances are creating unique opportunities to improve our ability to predict risk and assess the potential toxicity of substances in the products FDA regulates. "These cutting-edge technologies can potentially reduce or replace the use of mammals for toxicity testing," says Hunt.
Much of what we know about human biology and the fundamental properties of cells has come from studies of research organisms, often referred to as model organisms. A model organism is a species that has genetic similarities to humans and is widely used to research biological and disease processes because they have experimental advantages. Model organisms that are small, easy to breed in large numbers in a laboratory, and have a very short life cycle may provide rapid, human-relevant toxicity data at lower cost.
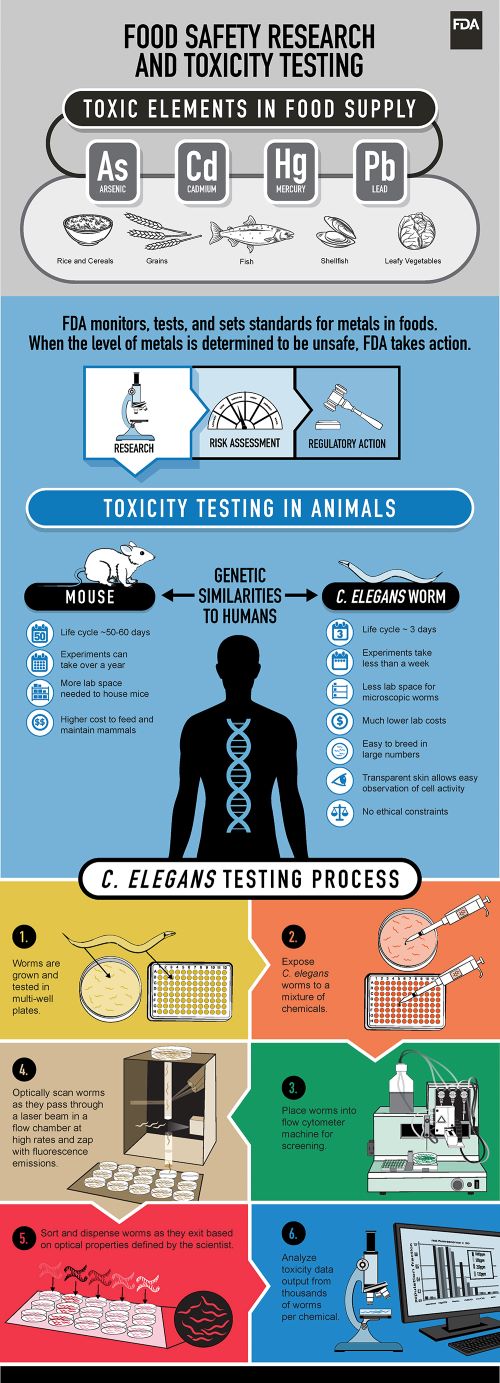
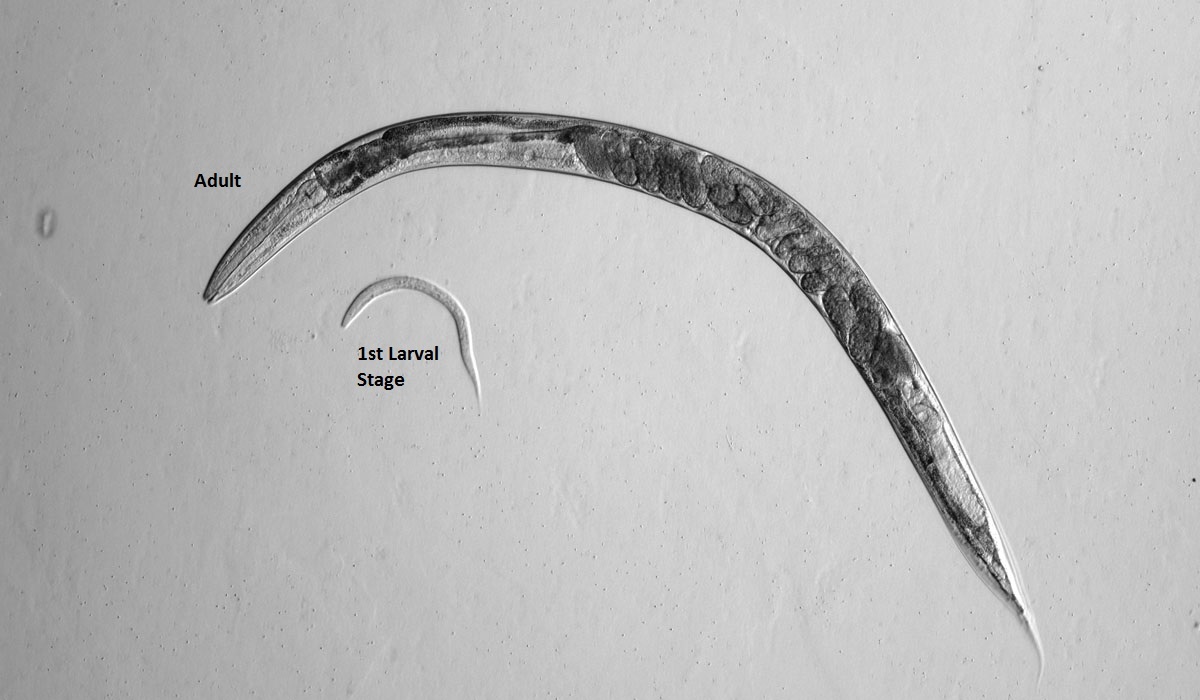
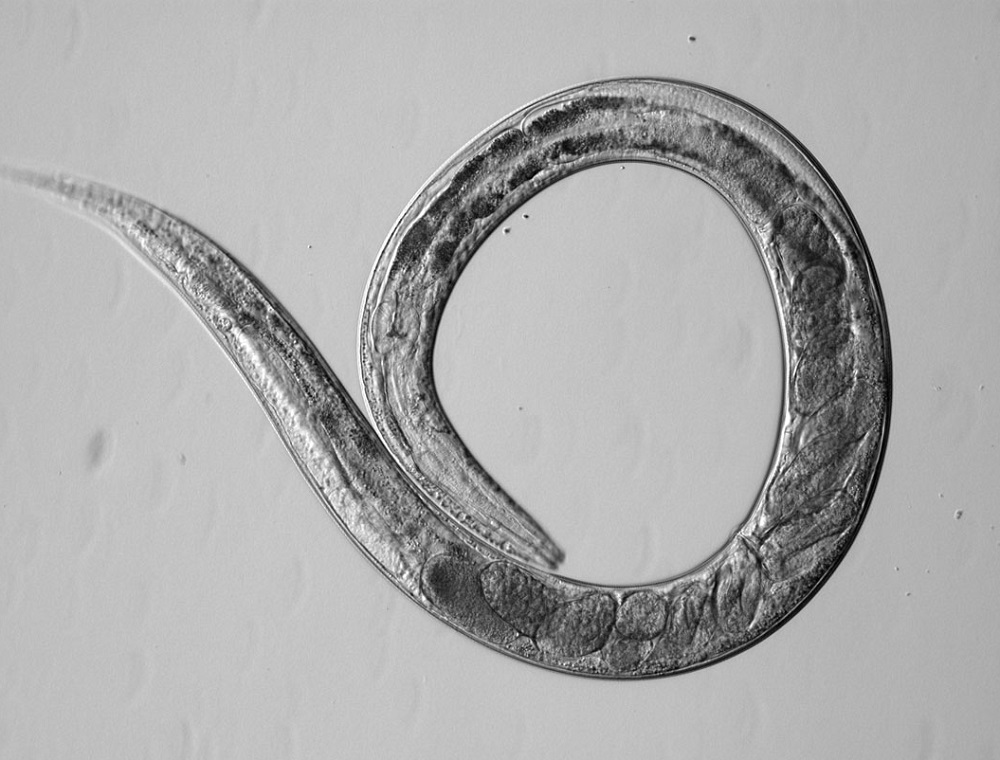
One of these model organisms now being tested by Hunt is Caenorhabditis elegans, a tiny transparent roundworm about 1 mm in length. It grows from egg to egg-laying adult in only 3 days and lives for about 3 weeks. Amazingly, the genes and molecular pathways of humans and worms are very similar. We often respond to toxins in similar ways.
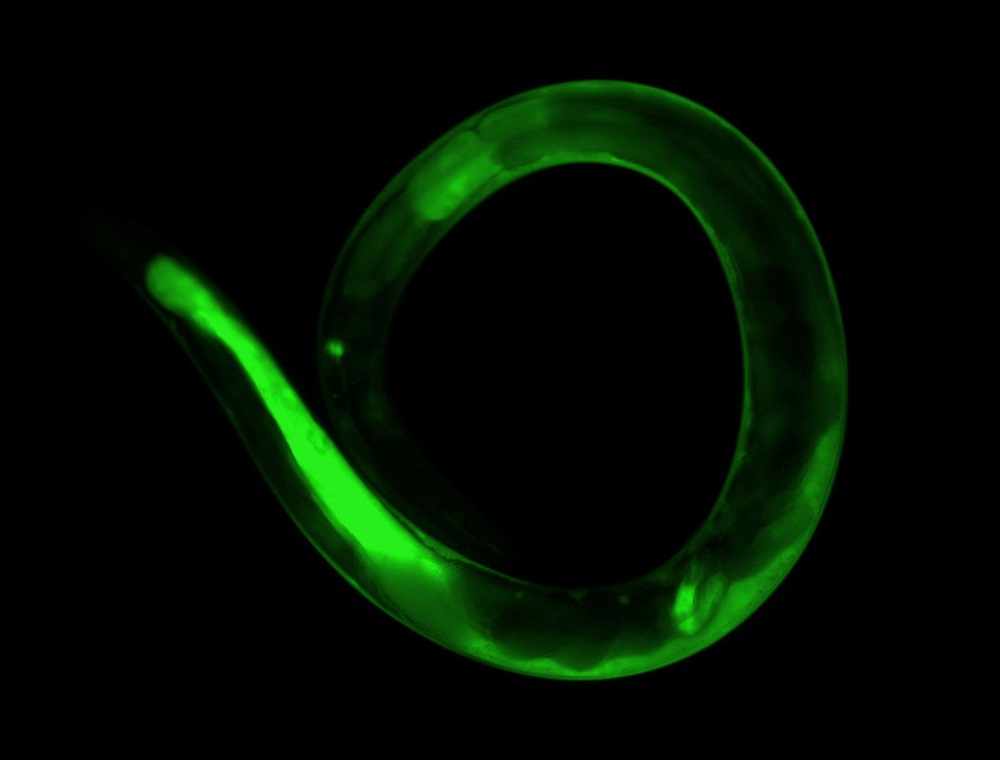
Because C. elegans are transparent, it's simple to trace the behavior of individual cells throughout its development as cells grow and move. Genetic modification can be easily observed in a living organism all the way from embryonic development to adulthood. Even though it doesn’t have a heart or blood, the simplicity of its body is what might make it a valuable mode.
Benefits of the Worm
According to Hunt, the C. elegans worm may have great potential to address the need for faster and more reliable testing methods for predictive toxicology. The duration and cost of running C. elegans tests is comparable to cell-based in vitro testing yet allows for toxic exposure information from a whole animal with intact and metabolically active digestive, reproductive, sensory, and neuromuscular systems.
Another major advantage of using C. elegans is laboratory logistics. Typically, experiments on rats can take over a year, whereas C. elegans testing takes less than a week. Within the space required to maintain 1 lab rat, you can fit over 200,000 C. elegans worms. They can be grown cheaply and in large numbers without expensive laboratory equipment. Moreover, healthy cultures of C. elegans can be frozen and then defrosted and revived when needed. Finally, the anatomy and development of C. elegans can be examined easily under a microscope.
Limitations of C. elegans
Although C. elegans has many biological similarities to humans, it also has major differences. These worms lack mammalian organs such as lungs, heart, kidney, and liver. They have an innate immune system but lack adaptive immunity.
At this time, any chemicals tested in C. elegans must be soluble in water, which limits the number of compounds that can be screened. However, Hunt’s lab is currently working on alternative solutions such as the use of nanoemulsions for testing fat soluble chemicals. Small changes in temperature, nutrient, or salt concentration elicit adaptive responses that can significantly alter analysis results, sometimes for multiple generations of the creature. That’s why good culture practice and specific handling procedures are so important.
Due to the limitations, Hunt and other scientists try to test their research findings in a variety of model organisms and must ultimately confirm the results through validation studies before C. elegans data can be used for regulatory decisions. Hunt has started to work with risk assessment experts to determine if C. elegans is fit for this purpose.
Promising Avenues for Toxicity Testing Using C. elegans
For now, toxicity testing in mammals will continue to be the standard used to predict safe human exposure levels of chemicals. However, no model is perfect, and even human trials do not always predict outcomes in the population at large.
Given the high percentage of available chemicals for which there is little or no toxicity data available, lower cost toxicity screens that can at least flag for further study those chemicals with the most potential for harm are urgently needed. As an intermediate between in vitro and mammal testing, chemical hazard ranking using C. elegans tests may predict toxicity ranking in mammals for developmental and acute toxins.
Facilities and Resources
The FDA laboratory where Hunt does her research has acquired complex, state-of-the-art equipment and software tools to conduct targeted toxicology testing.
One of these machines used for C. elegans analysis is an instrument that combines laser technology with microfluidics. It's like a flow cytometer designed to accommodate living multicellular specimens the size of C. elegans. The tool is used for high-throughput, automated analysis. It can optically analyze, sort, and dispense up to 100 worms per second on the basis of fluorescent protein expression patterns and other optical signatures. The worms pass through a focused laser beam in a flow cell at extremely high rates. This beam is narrower than the worm so that multiple measurements are made per animal, which means that the worm is optically scanned along its long axis. It's also sequentially zapped by fluorescence emissions from 3 separate fluorescent channels as the animal flows by.
The ability to dispense organisms as they emerge from the flow cell allows for the collection of those organisms that have certain optical properties defined by the scientist.
Hunt is developing a method to assess toxicity by measuring death in adult C. elegans using a dye as a fluorescent marker to identify worms that died from any cause. She analyzes the machine's data output from thousands of worms per condition and ranks compounds based on their known toxicity.
Conclusion
FDA toxicologists must make tough scientific decisions about the safety of the products we regulate. They are building the skills and experience to provide critical insights into the best ways to use these new technologies and incorporate them into regulatory review. FDA has developed a Predictive Toxicology Roadmap for integrating new toxicology methods into our safety and risk assessments. It's a big step in the right direction. If FDA accepts a scientific tool, such as the C. elegans testing model and others, industry can more easily use it during product development, saving everybody time and money.
Collaborators
- Piper Hunt, Ph.D.
- Robert Sprando, Ph.D.
- Jessica Camacho, Ph.D
- Bonnie Welch, M.S.