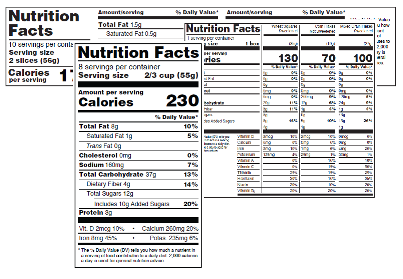
Changes to the Nutrition Facts Label
The Nutrition Facts label on packaged foods was updated in 2016 to reflect updated scientific information, including information about the link between diet and chronic diseases, such as obesity and heart disease. The updated label makes it easier for consumers to make better informed food choices. The updated label appears on the majority of food packages. Manufacturers with $10 million or more in annual sales were required to update their labels by January 1, 2020; manufacturers with less than $10 million in annual food sales were required to update their labels by January 1, 2021. Manufacturers of most single-ingredient sugars, such as honey and maple syrup, and certain cranberry products had until July 1, 2021 to make the changes.
Industry members, visit our For Industry page for additional resources, including questions and answers.
Consumers and educators, visit the Nutrition Facts Label Education Campaign page to learn more about the changes to the Nutrition Facts label.
See a Side-by-Side of the Original Label and New Label (PDF: 462KB), en Español (PDF: 463KB).
Highlights of the Updated Label
- The “iconic” look of the label remains, but we made important updates to ensure consumers have access to the information they need to make informed decisions about the foods they eat. These changes include increasing the type size for “Calories,” “servings per container,” and the “Serving size” declaration, and bolding the number of calories and the “Serving size” declaration to highlight this information.
- Manufacturers must declare the actual amount, in addition to percent Daily Value of vitamin D, calcium, iron and potassium. They can voluntarily declare the gram amount for other vitamins and minerals.
- The footnote now better explains what percent Daily Value means. It reads: “*The % Daily Value tells you how much a nutrient in a serving of food contributes to a daily diet. 2,000 calories a day is used for general nutrition advice.”
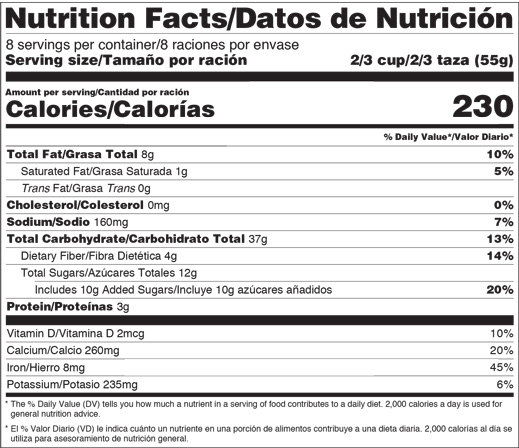
- “Added sugars,” in grams and as percent Daily Value, must be included on the label. There are different labeling requirements for single-ingredient sugars.
- The list of nutrients that are required or permitted to be declared is being updated. Vitamin D and potassium are required on the label. Calcium and iron will continue to be required. Vitamins A and C are no longer required but can be included on a voluntary basis.
- While continuing to require “Total Fat,” “Saturated Fat,” and “Trans Fat” on the label, “Calories from Fat” was removed because research shows the type of fat is more important than the amount.
- Daily values for nutrients like sodium, dietary fiber and vitamin D have been updated based on newer scientific evidence from the Institute of Medicine and other reports such as the 2015 Dietary Guidelines Advisory Committee Report, which was used in developing the 2015-2020 Dietary Guidelines for Americans. Daily values are reference amounts of nutrients to consume or not to exceed and are used to calculate the percent Daily Value (% DV) that manufacturers include on the label. The %DV helps consumers understand the nutrition information in the context of a total daily diet.
- By law, serving sizes must be based on amounts of foods and beverages that people are actually eating, not what they should be eating. How much people eat and drink has changed since the previous serving size requirements were published in 1993. For example, the reference amount used to set a serving of ice cream was previously 1/2 cup but is now 2/3 cup. The reference amount used to set a serving of soda changed from 8 ounces to 12 ounces.
- Package size affects what people eat. So for packages that are between one and two servings, such as a 20 ounce soda or a 15-ounce can of soup, the calories and other nutrients are required to be labeled as one serving because people typically consume it in one sitting.
- For certain products that are larger than a single serving but that could be consumed in one sitting or multiple sittings, manufacturers have to provide “dual column” labels to indicate the amount of calories and nutrients on both a “per serving” and “per package”/“per unit” basis. Examples would be a 24-ounce bottle of soda or a pint of ice cream. With dual-column labels available, people can more easily understand how many calories and nutrients they are getting if they eat or drink the entire package/unit at one time.
English (PDF: 955KB), Español (PDF: 1MB)
Resources and Additional Information
Final Rule: Revision of the Nutrition and Supplement Facts Labels
See submitted comments, supporting documents, and references in Docket No. FDA-2012-N-1210.
Final Rule: Serving Sizes of Foods That Can Reasonably Be Consumed At One Eating Occasion; Dual-Column Labeling; Updating, Modifying, and Establishing Certain Reference Amounts Customarily Consumed; Serving Size for Breath Mints; and Technical Amendments
See submitted comments, supporting documents, and references in Docket No. FDA-2004-N-0258.
Final Rule: Extension of the Compliance Dates for the Nutrition Facts and Supplement Facts Label and Serving Size Rules
See submitted comments, supporting documents, and references in Docket No. FDA-2012-N-1210 and Docket No. FDA-2004-N-0258.
1. Why did you change the Nutrition Facts label?
The previous label was more than 20 years old when the changes were made. To make sure consumers have access to more recent and accurate nutrition information about the foods they are eating, FDA required changes based on updated scientific information, new nutrition and public health research, more recent dietary recommendations from expert groups, and input from the public.
2. What major changes were made?
The changes include modifying the list of required nutrients that must be declared on the label, updating serving size requirements, and providing a refreshed design. The current Nutrition Facts label makes it easier for consumers to make informed decisions about the food they eat.
3. Is the updated label already on packaged foods?
Yes, manufacturers with $10 million or more in annual food sales had to make the changes by January 1, 2020. Manufacturers with less than $10 million in annual food sales had an extra year to comply – until January 1, 2021. Manufacturers of most single-ingredient sugars such as honey and maple syrup and certain cranberry products have until July 1, 2021 to make the changes.
4. Why must “added sugars” now be included?
The scientific evidence underlying the 2010, the 2015-2020, and the 2020-2025 Dietary Guidelines for Americans support reducing caloric intake from added sugars. Consuming too much added sugars can make it difficult to meet nutrient needs while staying within calorie limits.
The FDA recognizes that added sugars can be a part of a healthy dietary pattern. But if consumed in excess, it becomes more difficult to also eat foods with enough dietary fiber and essential vitamins and minerals and still stay within calorie limits. The updates to the label will help increase consumer awareness of the quantity of added sugars in foods. Consumers may or may not decide to reduce the consumption of certain foods with added sugars, based on their individual needs or preferences.
Sugars that are added during the processing of foods will have both the percent Daily Value and the number of grams of Added Sugars on their labels. Single-ingredient sugars such as table sugar, maple syrup, or honey will only have the percent Daily Value for Added Sugars listed on their labels. See the Nutrition Facts label for honey, maple syrup, or other single-ingredient sugars or syrups as well as for certain cranberry products.
5. Must Added Sugars be declared on packages and containers of single-ingredient sugars and syrups, such as pure honey and maple syrup?
Packages and containers of these products are not required to include a declaration of the number of grams of Added Sugars in a serving of the product but must still include a declaration of the percent Daily Value for Added Sugars. Manufacturers are encouraged, but not required, to use the “†” symbol immediately following the Added Sugars percent Daily Value on single-ingredient sugars, which would lead to a footnote explaining the amount of added sugars that one serving of the product contributes to the diet as well as the contribution of a serving of the product toward the percent Daily Value for added sugars.
6. What about certain cranberry products that have sugars added for palatability?
The number of grams of Added Sugars in a serving of a cranberry product, as well as the percent Daily Value for Added Sugars, must still be labeled. FDA intends to exercise enforcement discretion for certain cranberry products to allow manufacturers to use a symbol leading to a statement that is truthful and not misleading placed outside the Nutrition Facts label. These manufacturers could explain, for example, that the sugars added to certain dried cranberries or cranberry beverage products are added to improve the palatability of naturally tart cranberries. See the Nutrition Facts label for honey, maple syrup, or other single-ingredient sugars or syrups as well as for certain cranberry products.
7. How does the FDA define “added sugars”?
The definition of added sugars includes sugars that are either added during the processing of foods, or are packaged as such, and include sugars (free, mono- and disaccharides), sugars from syrups and honey, and sugars from concentrated fruit or vegetable juices that are in excess of what would be expected from the same volume of 100 percent fruit or vegetable juice of the same type. The definition excludes fruit or vegetable juice concentrated from 100 percent fruit juice that is sold to consumers (e.g. frozen 100 percent fruit juice concentrate) as well as some sugars found in fruit and vegetable juices, jellies, jams, preserves, and fruit spreads. However, although they are still “added sugars,” single-ingredient sugars such as pure honey, maple syrup, and a bag of sugar, have different labeling requirements from other added sugars.
For industry and those interested in the more technical version of the definition, please consult page 33980 of the Nutrition Facts Label Final Rule.
8. Are you using the new label to tell people what to eat?
The Nutrition Facts label is designed to provide information that can help consumers make informed choices about the food they purchase and consume. It is up to consumers to decide what is appropriate for them and their families’ needs and preferences.
9. Why is trans fat still on the label if the FDA is phasing it out?
Trans fat will be reduced but not eliminated from foods, so FDA will continue to require it on the label. In 2015, the FDA published a final determination that partially hydrogenated oils (PHOs), the source of artificial trans fat, are not generally recognized as safe, but this determination would not affect naturally occurring trans fat, which would still exist in the food supply. Trans fat is present naturally in food from some animals, mainly ruminants such as cows and goats. Also, industry can currently use some oils that are approved as food additives and can still petition FDA for certain uses of PHOs.
10. Why are vitamin D and potassium being added to the Nutrition Facts label?
Vitamin D and potassium are nutrients Americans don’t always get enough of, according to nationwide food consumption surveys (http://www.cdc.gov/nchs/nhanes/), and when lacking, are associated with increased risk of chronic disease. Vitamin D is important for its role in bone health, and potassium helps to lower blood pressure. Calcium and iron are already required and will continue to be on the label.
11. Why are you no longer requiring vitamins A and C?
In the early 1990’s, American diets lacked Vitamins A and C, but now Vitamins A and C deficiencies in the general population are rare. Manufacturers are still able to list these vitamins voluntarily.
12. Does the new label look different?
We have made some improvements to the format to provide significant public health information. Changes include:
- Highlighting “Calories,” “servings per container,” and the “Serving size” declaration by increasing the type size and placing the number of calories and the “Serving size” declaration in bold type.
- Requiring manufacturers to declare the actual amount, in addition to percent Daily Value, of the mandatory vitamins and minerals.
- Adding “Includes X g Added Sugars” directly beneath the listing for “Total Sugars.” Some sugars such as honey and maple syrup do not have to list the number of grams of added sugars but must still include the %Daily Value.
- Changing the footnote to better explain the percent Daily Value. It will now read: “*The % Daily Value tells you how much a nutrient in a serving of food contributes to a daily diet. 2,000 calories a day is used for general nutrition advice.”
13. I heard that some serving sizes are actually be bigger. That doesn’t seem to make sense with the obesity epidemic.
Some serving sizes will increase and others will decrease because by law, the serving sizes must be based on the amounts of food and drink that people typically consume, not on how much they should consume. Recent food consumption data show that some serving sizes need to be revised. For example, the reference amount used to set a serving of ice cream was previously ½ cup and now is 2/3 cup. The reference amount used to set a serving size of soda was previously 8 ounces and now is 12 ounces. The reference amount for yogurt decreased from 8 ounces to 6 ounces. Nutrient information on the new label will be based on these updated serving sizes so it matches what people actually consume.
14. Do the new requirements apply to imported food?
Yes, foods imported to the United States will need to meet the final requirements.
Format Examples
Download high-resolution examples of different labels in the new format (for reference only), includes the following label samples:
- Standard Vertical
- Standard Vertical (Side-by-Side Display)
- Standard Vertical (w/ Voluntary)
- Tabular Format
- Aggregate Display
- Dual Column Display
- Simplified Display
- Infants through 12 Months of Age
- Children 1-3 Years
- Tabular Dual Column Display
- Tabular Display for Small Packages
- Linear Display for Small Packages
- Dual Columns, Two Forms of the Same Food
- Dual Columns, Per Serving and Per Unit
- Declaration of Added Sugars for Single-Ingredient Sugars and Certain Cranberry Products (PDF 384 KB)
- Preliminary Regulatory Impact Analysis for the Proposed Rules on the Nutrition Facts Label
- Side-By-Side Comparison of the Old and New Nutrition Facts Label (PDF: 575KB) Español (PDF: 789KB)
- Details of Key Changes 1 Page Overview (PDF: 189KB) Español (PDF: 193KB)
- Infographic on Serving Sizes (PDF: 431KB) Español (PDF: 410KB)
- Examples of Labels in the New Format (PDF: 1.7MB)
- At a Glance: Highlights of the Final Nutrition Facts Label (PDF: 195KB)
On March 11, 2020, FDA launched its Nutrition Facts Label Education Campaign. “The New Nutrition Facts Label: What’s in it for You?” education campaign was developed by FDA to raise awareness about the changes to the Nutrition Facts label, increase its use, and help consumers, health care professionals, and educators learn how to use it as a tool for maintaining healthy dietary practices.
Learn More About
- What’s New with the Nutrition Facts Label
- Serving Size
- Calories
- Percent Daily Value
- Added Sugars
- Nutrition Facts Label and MyPlate