FDA Drug Safety Communication: Important safety changes to the influenza drug Tamiflu (oseltamivir phosphate) for oral suspension
Safety Announcement
Additional Information for Patients
Additional Information for Healthcare Professionals
Additional Information for Pharmacies
Additional Background Information (including product graphics)
[7-11-2011] The U.S. Food and Drug Administration (FDA) is informing the public of important product safety changes to the influenza drug Tamiflu (oseltamivir phosphate) for oral suspension. These changes are being made to reduce the possibility of prescribing and dosing confusion that can lead to medication errors. FDA has worked with the manufacturer, Genentech (part of the Roche Group), to make these changes.
The changes to Tamiflu oral suspension and the product label include:
Facts about Tamiflu
- In a class of medications called neuraminidase inhibitors. These drugs work by stopping the spread of the influenza (flu) virus in the body.
- Helps shorten the time you have flu symptoms such as a stuffy or runny nose, sore throat, cough, muscle or joint aches, tiredness, headache, fever, and chills
- Used to treat some types of flu in adults and children (older than 1 year of age) who have had symptoms for no longer than 2 days. It is also used to prevent some types of flu in adults and children (older than 1 year of age) when they have spent time with someone who has the flu or when there is a flu outbreak.1
- A change to the concentration of Tamiflu from 12 mg/mL to 6 mg/mL. The lower concentration of Tamiflu is less likely to become frothy when shaken, which helps to ensure an accurate measurement. The 12 mg/mL concentration will no longer be marketed after current supplies run out.
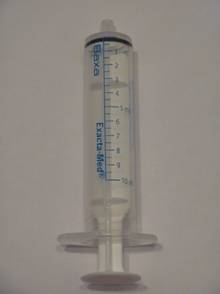
- A change in the measurements of the oral dosing device (graphic) from milligrams (mg = weight) to milliliters (mL = volume).
- A change in the dosing table for Tamiflu to include a column for the volume (mL) based on the new 6 mg/mL concentration. (Table 1)
-
Revised container labels and carton packaging (graphic).
-
Revised compounding instructions for pharmacies to prepare a 6 mg/mL oral suspension from Tamiflu capsules in an emergency situation only if the commercially manufactured Tamiflu for oral suspension is unavailable.
Genentech, the manufacturer of Tamiflu for oral suspension, plans to begin distribution of the new 6 mg/mL product in July 2011. The company has instituted a voluntary Take Back Program for wholesale buyers, distributors and pharmacies to remove the 12 mg/mL product from the marketplace.
There are no quality issues with the 12 mg/mL product—it is still useable through its expiration date. However, FDA encourages participation in the Take Back Program to limit the potential for product confusion.
The 12 mg/mL product will remain in the marketplace and in state or national stockpiles until current supplies expire. Therefore, it is important for healthcare professionals to be aware that a patient may potentially receive either concentration (6 mg/mL or 12 mg/mL) from their pharmacy during the next influenza season (2011-2012). Steps should be taken to avoid the potential for a medication error due to confusion between the two concentrations (see Additional Information sections below).
For complete background information on Tamiflu, please see Tamiflu (oseltamivir phosphate) Information.
Additional Information for Patients
- The concentration of Tamiflu for oral suspension is changing and you may get either concentration at your pharmacy during the next flu season.
- The new Tamiflu for oral suspension container label and carton packaging look different from what you may have taken in the past.
- The new oral dosing device is different and the volume (mL) of your dose may differ from past prescriptions.
- Check with a healthcare professional if you have any questions about the dosing directions, how to measure a dose using the new dosing device, or about which concentration of Tamiflu for oral suspension you have.
- Report any side effects you experience or medication errors to your healthcare professional and the FDA MedWatch program using the information in the “Contact Us” box at the bottom of the page.
Additional Information for Healthcare Professionals
- Prescribers should include the new concentration (6 mg/mL) and dose in milliliters on all prescriptions for Tamiflu for oral suspension. A revised dosing chart in Table 1 provides dosing in milliliters, and is included in the revised professional label.
- Prescribers should be aware that pediatric strength Tamiflu capsules (30 mg and 45 mg) are available and have not changed. These capsules can be prescribed for pediatric patients who can swallow capsules. For patients who can not swallow capsules, these can be opened and the capsule contents can be mixed with flavored foods (such as chocolate syrup or caramel topping).
- The new 6 mg/mL product contains an oral dosing device graduated in mL, whereas the old 12 mg/mL product contained an oral dosing device with dose markings in mg.
- It is possible that patients may get either concentration at the pharmacy during the 2011-2012 flu season; patients should be educated about this possibility to avoid medication errors.
- The two versions of the professional label may appear in circulation during the next influenza season (2011-12) and contain different dosing and compounding instructions for the oral suspension.
- Report adverse events or medication errors involving Tamiflu to the FDA MedWatch program, using the information in the “Contact Us” box at the bottom of the page.
Additional Information for Pharmacies
- The most important measure a pharmacy can take is to participate in the Take Back Program and replace the 12 mg/mL concentration of Tamiflu for oral suspension with the new 6 mg/mL concentration.
- Pharmacies should contact their distributor if they have unused/undispensed 12 mg/mL Tamiflu for oral suspension to determine if the product may be returned through the Take Back Program before August 31, 2011.
- Pharmacists should verify that each prescription for Tamiflu for oral suspension contains a concentration (mg/mL) and a dose (mL). If the concentration and dose are not specified, then the pharmacist should clarify the prescription with the prescriber.
- Pharmacists should ensure that the units of measure for the dosing instructions on the bottle match the oral dosing device that is provided (e.g., for the new 6 mg/mL product, the units of measure for both the drug and the device should be in milliliters).
- Pharmacists should ensure that the correct dose, dosing instructions, and oral dosing device are provided for the patient and are consistent with the concentration of Tamiflu for oral suspension (6 mg/mL or 12 mg/mL) that the patient will receive.
Additional Background Information
FDA has worked with Genentech to make these important product safety changes to the Tamiflu for oral suspension product and to address the medication error-prone issues that FDA previously identified and communicated.
FDA issued a Public Health Alert in September 2009 regarding the potential for medication errors with the use of Tamiflu for oral suspension. The agency had received reports of errors where dosing instructions for the patient did not match the dosing dispenser. Additionally, the commercial Tamiflu for oral suspension concentration (12 mg/mL) was different from the concentration of the compounded suspension for emergency use (15 mg/mL when made from Tamiflu 75 mg capsules), which also may have contributed to confusion and medication errors.
FDA issued a Public Health Advisory in December 2009 on the availability of Tamiflu for oral suspension during an overall product shortage. Because the concentration of the marketed Tamiflu for oral suspension has changed to 6 mg/mL, the emergency compounding instructions that FDA provided in 2009 are now being updated to provide a final concentration of 6 mg/mL. This change will align the emergency-compounded final concentration with the new commercially-marketed oral suspension concentration of 6 mg/mL, which may reduce the potential for prescribing and dosing errors. FDA reiterates that this compounded suspension should not be used merely for convenience, or whenthe FDA-approved Tamiflu for oral suspension available.
References
- U.S. National Library of Medicine. National Institutes of Health. Drug Monograph-Oseltamivir. Available at http://www.nlm.nih.gov/medlineplus/druginfo/meds/a699040.html. Accessed April 15, 2011.
New Tamiflu Dosing Chart (Table 1)
Table 1. Treatment and Prophylaxis Dosing of Oral Tamiflu for Influenza
For Patients 1 Year of Age and Older Based on Body Weight
|
Weight |
Weight |
Treatment |
Prophylaxis |
Volume of Dose* |
Number of |
Number of |
|---|---|---|---|---|---|---|
|
15 kg or less |
33 lbs or less |
30 mg twice daily |
30 mg once daily |
5 mL |
1 bottle |
10 Capsules 30 mg |
|
16 kg thru 23 kg |
34 lbs thru 51 lbs |
45 mg twice daily |
45 mg once daily |
7.5 mL |
2 bottles |
10 Capsules 45 mg |
|
24 kg thru 40 kg |
52 lbs thru 88 lbs |
60 mg twice daily |
60 mg once daily |
10 mL |
2 bottles |
20 Capsules 30 mg |
|
41 kg or more |
89 lbs or more |
75 mg twice daily |
75 mg once daily |
12.5 mL† |
3 bottles |
10 Capsules 75 mg |
* A 10 mL oral dosing dispenser is provided with the oral suspension. In the event that the dispenser provided is lost or damaged, another dosing dispenser may be used to deliver the volume.
†Delivery of this Tamiflu for oral suspension dose requires administering 10 mL followed by another 2.5 mL.
New Oral Dispenser/Syringe Device (10 mL) with markings in mL
New Container Label Photo (6mg/mL)
Old Container Label Photo (12 mg/mL)
Related Information
- Tamiflu (oseltamivir phosphate) Information
- Comunicado de seguridad de medicamentos de la FDA: Cambios importantes de seguridad del medicamento para la influenza Tamiflu (fosfato de oseltamivir) en suspensión oral
- FDA Drug Safety Podcast for Healthcare Professionals: Important safety changes to the influenza drug Tamiflu (oseltamivir phosphate) for oral suspension
- Concentration Lowered in Tamiflu Medication
- FDA Public Health Alert: Potential Medication Errors with Tamiflu for Oral Suspension
- Public Health Advisory: Availability of Tamiflu for Oral Suspension