Reflections on a Landmark Year for Medical Product Innovation and Public Health Advances and Looking Ahead to Policy in 2018
January 9, 2018
By: Scott Gottlieb, M.D.

FDA Commissioner Scott Gottlieb, MD
As we look ahead to 2018, I’d like to take a moment to reflect on an inspiring year of advances in both medicine and public health for FDA — from groundbreaking medical products brought to market this year, to a record number of generic drug approvals that will promote competition, and to the agency’s ongoing efforts to advance policies that promote safe and effective product innovation, and keep Americans safe from food-related illnesses.
Today, new medical breakthroughs are profoundly altering how we view and treat disease in ways that seemed inconceivable just years ago. In this modern medical setting, FDA is evaluating all aspects of its policies to make sure we’re protecting consumers, while promoting beneficial innovation that has the potential to effectively treat disease for human and animal patients, and improve public health.
A Record Year for New Innovation
As scientific understanding of disease advances and the practice of medicine becomes more tailored to individual patient needs, we also are modernizing how we work with innovators throughout the development process to bring products to patients more efficiently, using the best available science.
For example, FDA recently coordinated the approval of a novel diagnostic device that can detect hundreds of genetic mutations in a single test with the Centers for Medicare & Medicaid Services’ proposed coverage of the test, thereby facilitating earlier access to this innovative product.
Also, in the rapidly advancing field of individualized medicine, the Agency advanced new draft guidance that addresses better ways to develop treatments that address the underlying molecular changes (e.g., genetic mutations) that often cause or contribute to diseases. This includes uncommon molecular changes that are present in only a small subset of patients. The guidance proposes an approach for drug developers to enroll patients into clinical trials for targeted therapies based on the identification of rare mutations, when reasonable scientific evidence suggests the drug could be effective in patients with these genomic findings. The new guidance discusses the evidence needed to demonstrate effectiveness for a variety of molecular subsets within a particular disease. The framework could lead to more consistent development and approval of targeted therapies for patients who are likely to benefit from them.
This past August we also saw the practical advent of a whole new way to treat disease with the approval of the first gene therapy product in the United States. We have since approved two more gene therapy medicines. Innovations like these are creating a turning point in the treatment of serious illnesses. With this technology also comes greater potential to cure intractable and inherited diseases.
2017 saw a number of other similar, historic milestones with regard to new innovation. This collective progress reflects a fundamental shift in science that’s enabling us to attack more diseases with novel platforms. We’re increasingly able to identify patient benefit earlier in the development process because of the ability to better target medicines to the underlying mechanisms of disease. At the same time, in many cases these identical tools also allow us to surface safety issues earlier and more effectively.
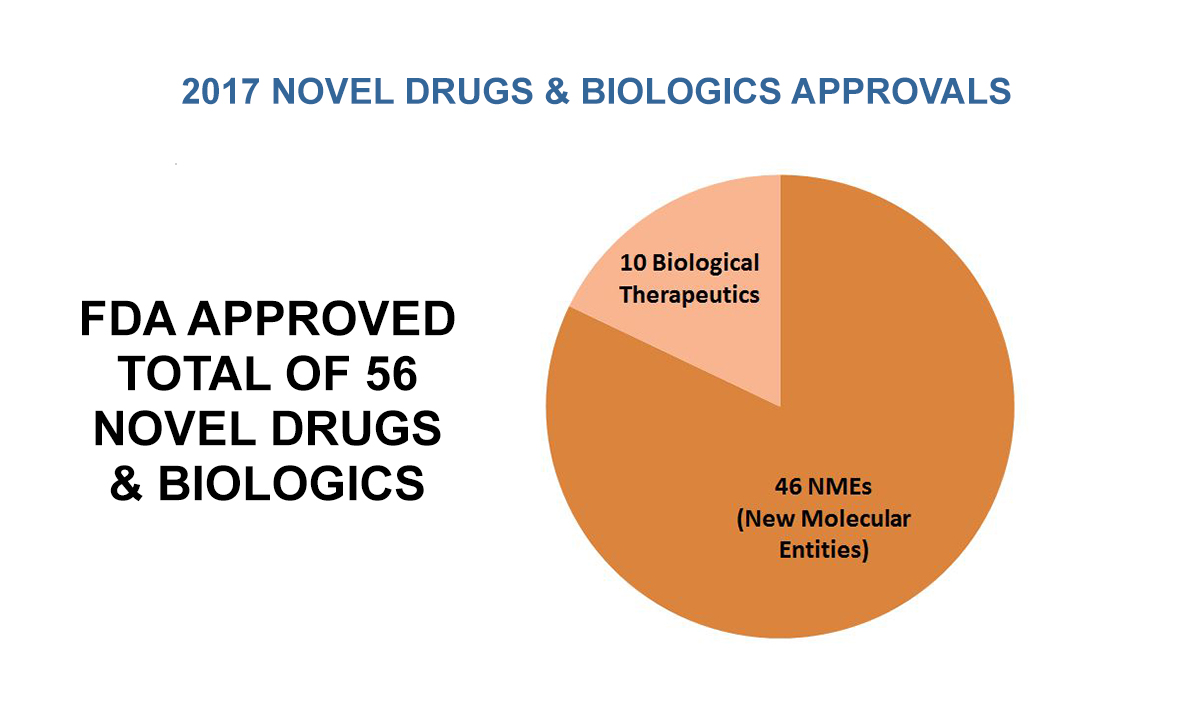
Owing in part to these advances, FDA approved a modern record number (56) of novel drugs and biologics in 2017. Of these 56 novel approvals this past year, 46 were new molecular entities approved by our Center for Drug Evaluation and Research – of which 28 were approved using one or more of FDA’s expedited review programs. Ten of these 56 novel approvals were biological therapeutics that were approved by our Center for Biologics Evaluation and Research. We also had a record number of drugs with orphan indications approved. At the same time we eliminated the entire backlog of pending orphan drug designation requests. We also broke records, with the highest number of generic drugs approved in a single month multiple times in 2017, and we recorded the highest annual total of generic drug approvals (1,027) in the agency’s history. We believe that, if current trends continue, we’ll exceed this record number of generic drug approvals in 2018.
Both full approvals and tentative approvals, which do not allow the applicant to market the generic drug product and postpones the final approval until all patents/exclusivity issues have expired.
Our science-based and patient-centered regulatory approach also extends to medical devices, where we’ve focused on a life-cycle approach to product development. This has allowed us to streamline clinical development protocols without compromising on our commitment to rely on rigorous evidence. By carefully considering when clinical data can be better gathered through post-market, as opposed to pre-market, studies, patients are waiting less time to access some breakthrough devices without conceding one bit FDA’s gold standard for demonstrating reasonable assurance of device safety and effectiveness.
In 2017, the agency approved a record number of novel devices — 95. This was more than four times the number of novel devices that received market approval in 2009.
Our commitment to applying the “least burdensome standard” for generating information critical for device approval was strengthened and advanced by provisions in the 21st Century Cures Act. This policy approach is a hallmark of our efforts to help innovators generate high quality evidence that can support marketing approval as efficiently as possible. Our embrace of these principles has resulted in remarkable advances in access for patients. In 2017, the agency approved a record number of novel devices — 95. This was more than four times the number of novel devices that received market approval in 2009.
Modernizing FDA’s Regulatory Programs
These new advances also present new challenges. At FDA, we’re being confronted with the need to regulate highly novel areas of science like gene therapy, targeted medicine, cell-based regenerative medicine, and digital health; where our traditional approaches to product regulation may not be as well suited. To meet these new challenges, we’re taking a fresh look at how we can adapt our customary approaches to regulation. We need to make sure that we’re allowing beneficial new technologies to advance, while continuing to protect consumers as part of our product review processes.
To promote these efforts, we advanced a new policy framework allowing certain diagnostic tests to undergo review by accredited third parties. This new framework will reduce the burden on test developers and streamline the regulatory assessment of these types of innovative products. This approach more readily accommodates the highly iterative nature of these technologies, where tests often undergo routine modifications to improve their precision and clinical utility.
Over the past summer, we also launched a pilot program exploring a new way of regulating digital health devices so that these fast-evolving technologies can similarly undergo the rapid product evolution that’s the hallmark of software tools like medical apps, while FDA maintains the ability to make sure that these digital health tools are being reliably produced. We followed these actions with a suite of guidances that clarify how we intend to regulate certain digital health technologies in a way that encourages innovation.
More broadly, we provided more clarity for manufacturers of low- to moderate-risk medical devices which will reduce unnecessary submissions to FDA for minor modifications that could not significantly affect device safety or effectiveness. As a result, patients will benefit from upgraded products more quickly.
This effort to properly match our policies to the unique attributes of the new technologies we’re being asked to review was also evident in new steps we took across other programs; from our comprehensive policy on regenerative medicine aimed at spurring safe and effective innovation in these potentially transformative products, to our draft guidance for manufacturers of 3D printed medical devices.
We know that the public health benefits derived from our efforts to modernize our regulatory approaches are not confined to the pre-market review process. Advances in our post-market tools and policies can yield meaningful advances for patients in the form of safer products, better information to guide medical decisions, and more opportunity to more efficiently move products to market – if we can have confidence in our post-market oversight. This is why we’re always looking for ways to reform and improve this oversight, and advance the ways that we share this information with patients and providers.
For example, last fall we launched a new searchable database to better inform patients and health care professionals of adverse events reported with drug and biologic products. We’ll be taking other steps soon to improve on the ways that we share important clinical information with patients and providers. These goals also include new efforts to step up our post-market oversight of potentially risky products, and warn consumers earlier of potential problems we find. As an example, we’ve taken decisive action to protect the public from risky stem cell products offered by unscrupulous clinics. We’ll pursue similar actions in 2018.
In 2017, we also took new steps to warn companies making false claims that their unapproved products can treat or cure life-threatening diseases; we advanced a new draft guidance describing FDA’s approach to regulating homeopathic products based on the risk they can pose to consumers; and we took steps to alert the public to the dangers of other unproven and untested products, such as certain body-building products, contaminated dietary supplements and kratom. Among other efforts, we also took new steps to facilitate faster patient access to needed compounded medicines, while protecting the public from poorly compounded drugs. There will be additional enforcement steps in 2018. And we continue to promote work that will enable FDA to use real world data to better inform our regulatory decision-making.
Promoting Drug Competition
Many say that FDA has no role in drug pricing, but I disagree. While we don’t have the authority to regulate prices, we do have the authority — and the responsibility — to ensure that the agency’s policies are not impeding competition that could ultimately be a check to rising drug prices and patient access.
Our role as gatekeeper of cost-effective, high-quality generic drug products is a foundational part of fostering human and animal drug competition. We’re advancing new ways that FDA can help enable patients to get access to more affordable medications. We shared some of the steps the agency is taking with our launch in June 2017 of the Drug Competition Action Plan — from prioritizing our review of generic drug applications, to working to stop companies from finding loopholes in the system that delay the entry of generic drugs to market, to making substantial progress on the generic drug review backlog, to ensuring that low cost drugs get to the patients who can benefit from their effectiveness and more affordable price.
New Steps to Combat Addiction
I’ve noted many times that among my highest priorities as Commissioner is addressing addiction crises facing the nation, principally with respect to nicotine and opioids. In 2017 we announced new plans for how we address these crises. In July, we announced a comprehensive plan that proposes to lower nicotine in combustible cigarettes to minimally or non-addictive levels. At the same time, we took new steps to enable development of innovative delivery systems that could be potentially less harmful than cigarettes for adults who still want to get access to satisfying levels of nicotine. As part of that plan, we formed a new Nicotine Steering Committee. It’s charged with modernizing FDA’s approach to development and regulation of nicotine replacement therapy products that can help smokers quit and stay quit
FDA also unveiled new actions to confront the staggering human and economic toll created by opioid abuse and addiction, starting with my first major action as commissioner to establish an Opioids Policy Steering Committee. Under the leadership of this committee, FDA is reevaluating how drugs that are already on the market are used, both for legitimate purposes and misuse and abuse.
The committee will recommend new policy steps to address this crisis. FDA also is taking immediate action where needed, as we did with FDA’s first-of-its-kind request to remove a currently marketed opioid pain drug from sale due to the public health consequences associated with the product’s abuse and misuse. We’ve also worked to identify ways to decrease exposure to opioids, prevent new addiction, and support the treatment of those with opioid use disorder; for example, through new Risk Evaluation and Mitigation Strategy requirements for makers of immediate-release opioids, and requiring labeling changes to add important clarifying information regarding the use of medication-assisted treatments for patients suffering from opioid use disorder. We’re continuing to pursue other creative ways to address the crisis, such as leveraging different forms of packaging, storage and disposal of opioid medications.
Protecting and Empowering Consumers
It’s not only medical products and policies where FDA can innovate to better serve the public – we’ve also made much progress in the implementation of the Food Safety Modernization Act, which was designed to keep the American public safe from food-related illness. Implementing the most comprehensive food safety reform in 70 years requires a massive commitment from federal, state and local governments to food producers, farmers and other stakeholders that are working to protect the public health in new and innovative ways. That’s why in July we announced more than $30 million in funding for states to help implement new produce safety requirements. We also launched an innovative software tool called the Food Safety Plan Builder that assists food manufacturers in creating a food safety plan to help prevent foodborne contamination and ultimately protect public health. And we’re looking at other ways to empower farmers and producers to ensure the law’s modernized requirements are effectively fulfilled.
I also believe in empowering people to make better choices. This is reflected in our continued efforts to pursue the practical implementation of the menu-labeling rule. We listened to public feedback and have proposed practical solutions to make it easier for industry to meet obligations in these important public health endeavors, while ensuring restaurant patrons have access to the nutrition information they need.
Empowerment also is critical for patients facing life-threatening or debilitating illness. This year we held our first-ever patient engagement advisory committee meeting. This is a pioneering effort that seeks to strengthen our engagement with patients and secure the patient voice in our regulatory decision-making.
We also understand that in many serious diseases, patients want earlier access to experimental treatments. We’ve taken new steps to improve the expanded access resources we have to serve patients, including enhancing our online Expanded Access Navigator Tool and simplifying the process for approving a patient’s request for access to an investigational treatment. Last month, as part of our commitment to expediting drug development for rare diseases, we issued draft guidance that describes a possible new approach for companies to collaborate and test multiple drug products in the same clinical trials for a specific ultra-rare pediatric disease, thereby reducing the number of patients that need to be treated with placebo. This framework can be applied more widely to other ultra-rare diseases.
In everything we do at FDA, our top priority is to protect the public health. Perhaps nowhere was this more evident in 2017 than in the areas of the United States that were impacted by last year’s hurricanes.
The devastation caused by Hurricanes Harvey, Maria and Irma brought to public view some of the critical work FDA does in overseeing the safety of the food and medical products. We worked around the clock to ensure that farmers in Texas and Florida could safely handle their crops affected by flooding. We remain deeply committed to the recovery in Puerto Rico and that island’s long-term success. We worked closely with drug and medical device manufacturers in Puerto Rico to take steps to address potential and apparent shortages of medical products that resulted from the devastation left by Hurricane Maria.
Although we’re seeing progress in Puerto Rico owing to the hard work of federal and local authorities — and primarily because of the resilience of the American people who are affected — our work and commitment to hurricane victims and patients in need of critical medical products will continue into 2018.
Improving our Stewardship of Vital Drugs
Antimicrobial resistance continues to be a major public health challenge. Addressing this problem requires adoption of a “one health” approach that involves new efforts to use antibiotics more responsibly in both human and animal medicine. To better manage antibiotic usage in food animals, in 2017 FDA completed its implementation of a groundbreaking Guidance for Industry (GFI #213). This new guidance document eliminates the use of medically important antimicrobials for food production purposes and brings the remaining therapeutic uses of antibiotics in animals under veterinary oversight.
A total of 292 approved drug applications were impacted by this new guidance; with 84 drug applications withdrawn, 93 applications for oral dosage form products intended for use in water converted from over-the counter (OTC) to prescription, and another 115 applications for products intended for use in feed converted from OTC to Veterinary Feed Directive. This is a major accomplishment. It represents a milestone in our efforts to promote judicious use of antibiotics in animal health. We will take additional steps in 2018 to build on these successes, and improve stewardship over medically important antibiotics.
A look ahead to 2018
When I look back at my eight months as Commissioner, since coming aboard at FDA in May, I’m humbled by the many accomplishments of the agency’s dedicated professional staff.
We’ve achieved a great deal in 2017. We’re committed to making even more progress this year. 2018 holds promise in even more areas where the agency will take steps to advance beneficial innovation by adopting new measures to make sure our processes are efficient, human and animal products are safe, and practical solutions are implemented that protect and promote the public health. The launch of our Unified Agenda highlights some of our priorities. These include advancing biosimilar policies, modernizing how we advance over-the-counter products, and better informing women about health issues and risk factors.
Nobody innovates in a silo. Advancements in medicine, biotechnology, food science, and the whole of public health are possible only because of the collaboration of the public health community. FDA is in a unique position to bring together stakeholders from across the sector — patients, industry, academics, providers, other government agencies — to ensure innovation translates into successful outcomes that protect and benefit the public. That’s what drives us at FDA. It’s what we’ll pursue in the year ahead.
Scott Gottlieb, M.D., is Commissioner of the U.S. Food and Drug Administration