2017 Was Another Record-Setting Year for Generic Drugs
January 17, 2018
By: Kathleen “Cook” Uhl, M.D.

In 2017, FDA’s Center for Drug Evaluation and Research’s generic drug program marked several major accomplishments on behalf of the American people.
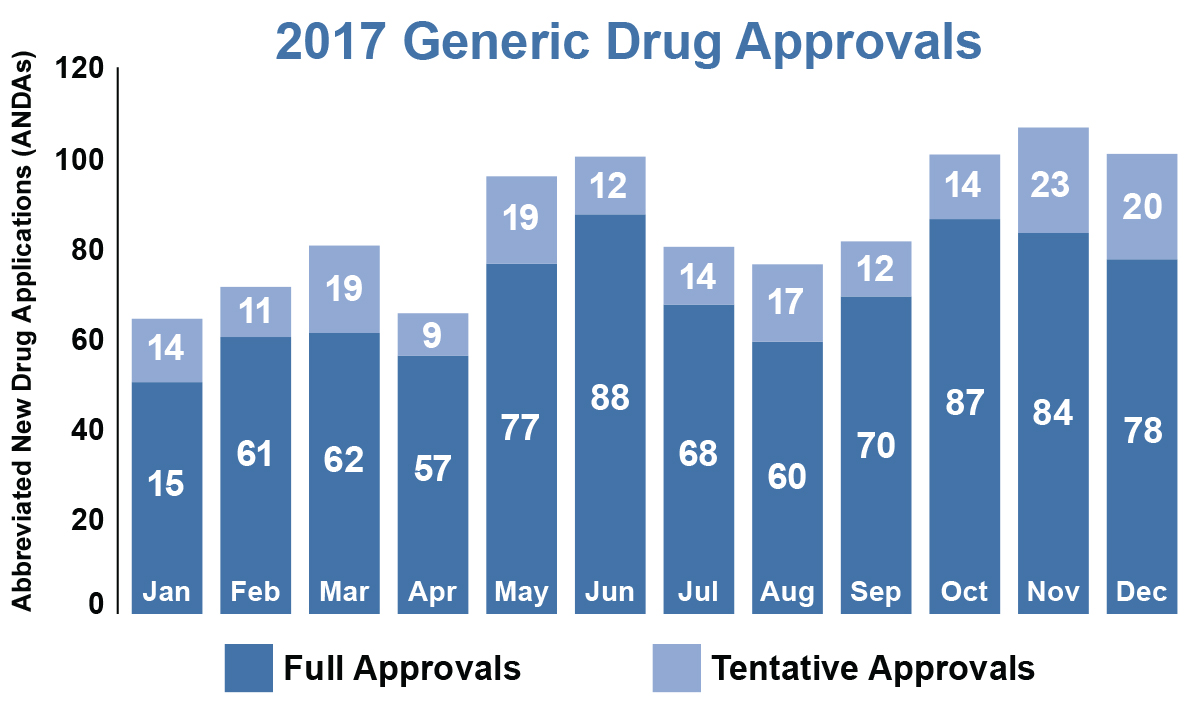
Our Office of Generic Drugs (OGD) marked another record-setting year for generic approvals at FDA with 1,027 new generic drugs, 214 more than our previous record of 813 set in 2016. Of those, 843 were full approvals and 184 were “tentative” approvals, that is, applications that are ready for approval from a scientific perspective, but cannot be fully approved due to patents or exclusivities on the brand-name drug.
Also in 2017, we helped establish the first reauthorization of the Generic Drug User Fee Amendments (known as GDUFA II) ̶ an important law that Congress passed to authorize the continued collection of user fees from generic drug manufacturers. GDUFA I, enacted in 2012, allowed OGD to hire additional staff, so that from 2012 to 2017 FDA had additional resources to approve the record numbers of generic drug applications. Reauthorization is helping facilitate continued advances in generic drugs, including complex drug products – such as some inhaled or injectable products.
FDA-approved generic drugs account for 89% of the prescriptions dispensed in the United States. Over the last decade, these FDA-approved generic drugs have saved consumers more than $1.67 trillion. While it’s exciting to see the number of approvals continue to rise year after year, and to exceed 1,000 annual approval actions for the first time, our attention remains focused on public health by ensuring the effectiveness and quality of approved generic drugs.
In 2017, we approved 80 “first generic” drugs. These are the first generic alternatives to a brand-name product. First generic drugs spur cost-saving competition that helps lower prescription drug costs. Lowering the cost of drugs is a public health priority, so FDA expedites the review of first generic applications to open the market to generic competition. In addition, multiple generic versions of the same drug lead to more competition, resulting in even more cost savings. In 2017, we updated our policy to prioritize the review of generic applications up to the third generic approval of a drug, helping to maximize savings for the public.
Another FDA initiative designed to foster competition focuses on complex drugs. OGD’s regulatory science work and guidances helped advance scientific knowledge about generic drugs to assist industry. OGD’s efforts provide the critical information needed to develop and meet our standards for equivalence to the brand-name drug. But traditional methods and standards for assessing generic drugs may not apply to more complex generics. Health care professionals use complex drugs to treat a wide range of diseases, from hormone replacement therapy in post-menopausal women to type II diabetes. In 2017, OGD provided guidance to industry on developing products from tiotropium bromide inhalation powder (the generic of Spiriva Handihaler), used to treat COPD, to EpiPen (epinephrine) alternative Adrenaclick, used for emergency treatment of anaphylaxis.
It’s important to note that even as FDA continues to meet the GDUFA performance goals, there will be occasional variations in generic drug approvals. Approval numbers can be impacted by a number of external factors, including the number of ANDAs submitted for review over a given time period and changes in legal requirements that come into effect that generic applicants must address to meet the standards for approval.
FDA’s continued work under GDUFA II will help ensure that safe and effective generic versions of brand-name drugs continue to be made available by giving industry clear guidelines on the science behind developing a quality generic drug and clearly identifying what is needed in an application to make it approvable.
The 2017 annual report provides more details on how OGD’s work benefits public health. We look forward to continuing our work with industry, the research community, physicians, health care providers, lawmakers, and other stakeholders to make generic drugs available for the benefit of the American public.
Kathleen “Cook” Uhl, M.D., is FDA’s Director, Office of Generic Drugs in the Center for Drug Evaluation and Research