Impact Story: Finding a Better Test for Predicting the Risk Drugs Pose to the Heart
FDA research has laid the groundwork for replacing the thorough QT (TQT) study, a costly clinical trial required to determine whether new drugs cause abnormal heart rhythms (arrhythmias), with a more precise, less costly, and faster approach to testing. Drugs with a high risk of causing arrhythmias will continue to be screened out, while potentially safe and effective drugs that might not have passed the TQT study will continue in development.
Several drugs were removed from the market in the 1990s and early 2000s because of un- or under-appreciated risk of a fatal arrhythmia. Beginning in 2005, most new drug compounds delivered through the bloodstream needed to pass a separate clinical trial, in addition to the traditional (Phases I-III) clinical trials that FDA required for drug development. This additional trial, called the TQT study, was added to determine whether the drug could cause a fatal heart arrhythmia.
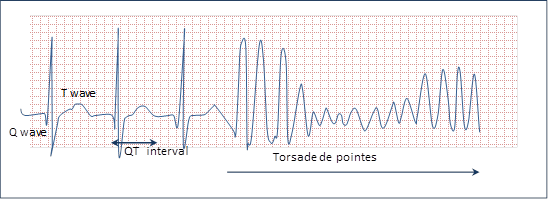
In a TQT study, healthy study participants are given a high dose of a candidate drug, and their heart activity is recorded on an electrocardiogram (ECG). If the drug prolongs the average time between the Q and T waves on the ECG more than 10 milliseconds, additional patient monitoring is required in clinical studies and development of the drug may stop.
Since 2005, approximately 450 TQT studies have been performed, costing drug developers more than $1 billion. No new drugs have been removed from the market because of arrhythmia risk since the test became mandatory, but it is not clear that the TQT approach identifies only the drugs with true risk.
How Did the TQT Study Come to Be Required for New Drug Compounds?
In the 1950s, doctors noted that some heart patients treated with quinidine developed fainting spells, convulsions, and, in some cases, sudden cardiac death. ECGs of these patients showed a rapid abnormal heartbeat that produced a spiked pattern called “torsade de pointes” (twisting of the points).
On the ECG, torsade was preceded by a subtle change, an increase in the QT interval—the time between the start of the Q wave and the end of the T wave in the heart's electrical cycle.
Drugs shown to prolong the QT interval and implicated in torsade included antihistamines, antidepressants, chemotherapeutics, pain medications, and others. Several of these drugs were withdrawn from the market. As the link between QT prolongation and potentially fatal cases of torsade became clearer, FDA began requiring a separate TQT study for most new drug molecules.
How Do Drugs Cause Harmful Arrhythmia?
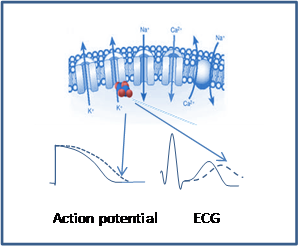
The heartbeat is controlled by an electrical signal that passes through the heart (the ECG records this signal from various locations on the skin). In the individual muscle cells (myocytes) of the heart, this signal is caused by the opening and closing of specialized channels in the membrane enclosing the cell that control the passage of calcium, sodium, and potassium ions.
The movement of these ions across the cell membrane causes a cyclic electrical change known as the action potential, which signals the muscle cell to contract. Action potentials are quickly propagated, so an electrical current and a wave of contraction passes through the heart muscle in the course of a heartbeat.
When a drug interferes with one or more kinds of ion channels, the action potential and the electrical current that controls the heart's contraction may be disrupted; this can lead to dangerous arrhythmias.
FDA and Partners Identify TQT Study Alternatives
Streamlining Clinical Evaluation by Incorporating the TQT Study Into Phase I Trials
Recognizing the costs and additional time required for the TQT study, FDA and its scientific collaborators wanted to know if it was possible to measure QT prolongation in the Phase I study (i.e., in a small number of people being given escalating doses of a drug) instead of in a separate TQT study. FDA and its collaborators conducted a study designed like a Phase I trial of drugs that had already undergone a TQT study and used an analytical approach called exposure-response modeling to determine QT prolongation. The results were consistent with the drug-induced changes in QT intervals observed in the previous TQT studies, demonstrating that ECG assessment in Phase I studies was an appropriate replacement for a TQT study.
Based on this study and other findings, FDA, along with international regulatory agencies acting through the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use, have revised their regulatory guidelines and no longer require a separate, dedicated TQT study for most new drugs, but some safe drugs are probably still being considered risky.
Using ECG Data to Develop More Specific Biomarkers for Heart Arrhythmia
Many drugs that prolong the QT interval do not cause a fatal arrhythmia, because of how they interact with ion channels that govern the passage of calcium, potassium, and sodium ions across the cell membrane. Although interaction with a particular potassium ion channel called hERG is a known cause of QT interval prolongation and arrhythmia, a drug that simultaneously affects sodium and calcium channels may actually stabilize heart rhythm, even though the QT interval is prolonged. Knowing this, CDER researchers have identified new measures based on the ECG waveform that may be better biomarkers of risk for torsade and subsequent arrhythmias. Read an article about this research: “Late sodium current block for drug-induced long QT syndrome: Results from a prospective clinical trial.”
CiPA: Developing a Better Paradigm for Predicting Risk of Arrhythmia Posed by New Drugs
Accurately predicting a drug's effect on the heart means understanding the drug's effects on each of the different types of ion channels and how these effects combine to influence the electrical activity of the cell and the heart as a whole. FDA is leading a global collaborative effort to create the Comprehensive In vitro Proarrhythmia Assay (CiPA). ![]()
CiPA is a risk prediction tool based on in vitro measurements of a drug's effect on each channel type in the heart's muscle cells. When combined with early-phase ECG testing using the exposure-response approach and validated with more specific ECG-based biomarkers, the CiPA tool would constitute a new regulatory approach that could greatly reduce the attrition of potentially beneficial drugs.
A 2017 FDA Advisory Committee endorsed the approach being taken by CiPA to validate a new tool for risk assessment of new drugs.
Learn more about the CiPA initiative. ![]()
For more information, please visit the Office of Translational Sciences.
Related Publications
- Sager PT, Gintant G, Turner JR, et al. Rechanneling the cardiac proarrhythmia safety paradigm: a meeting report from the Cardiac Safety Research Consortium. Am Heart J. 2014;167(3):292-300. This white paper describes the motivation and scientific thinking behind current efforts to develop a new paradigm for assessing cardiotoxic risk using nonclinical in vitro models.
- Johannesen L, Vicente J, Mason JW, et al. Late sodium current block for drug-induced long QT syndrome: Results from a prospective clinical trial. Clin Pharmacol Ther. 2016;99(2):214-23. This clinical study provides insight on how drugs that affect sodium channels in the heart can alter the QT interval and mitigate risk.
- Johannesen L, Vicente J, Mason JW, et al. Differentiating drug-induced multichannel block on the electrocardiogram: Randomized study of dofetilide, quinidine, ranolazine, and verapamil. Clin Pharmacol Ther. 2014;96(5):549-58. This clinical study provides new insights into how drug interactions with individual ion channels in the heart can affect heart function as revealed on an ECG.
- Darpo B, Benson C, Dota C, et al. Results from the IQ-CSRC prospective study support replacement of the thorough QT study by QT assessment in the early clinical phase. Clin Pharmacol Ther. 2015;97(4):326-35. This clinical study provides evidence that QT assessment in standard early-phase tolerability and dose-finding studies could replace the thorough QT study.
- Colatsky T, Fermini B, Gintant G, et al. The Comprehensive in Vitro Proarrhythmia Assay (CiPA) initiative—Update on progress.
--]) J Pharmacol Toxicol Methods. 2016;81:15-20.
J Pharmacol Toxicol Methods. 2016;81:15-20. - Vicente J, Stockbridge N, Strauss DG. Evolving regulatory paradigm for proarrhythmic risk assessment for new drugs.
--]) J Electrocardiol. 2016;49(6): 837-842.
J Electrocardiol. 2016;49(6): 837-842.
Related Guidance
Guidance for Industry: E14 Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs (PDF - 97.8 KB)