Drug Trials Snapshot: LONSURF
HOW TO USE THIS SNAPSHOT
The information provided in Snapshots highlights who participated in the clinical trials that supported the FDA approval of this drug, and whether there were differences among sex, race and age groups. The “MORE INFO” bar shows more detailed, technical content for each section. The Snapshot is intended as one tool for consumers to use when discussing the risks and benefits of the drugs.
LIMITATIONS OF THIS SNAPSHOT:
Do not rely on Snapshots to make decisions regarding medical care. Always speak to your health provider about the risks and benefits of a drug. Refer to the LONSURF Prescribing Information for complete information.
LONSURF (trifluridine and tipiracil)
lawn-surf
Taiho Oncology, Inc.
Approval date: September 22, 2015
DRUG TRIALS SNAPSHOT SUMMARY:
What is the drug for?
LONSURF is a drug for the treatment of metastatic colorectal cancer (cancer of the colon or rectum) that has spread to other parts of the body for patients who are no longer responding to other therapies.
How is this drug used?
LONSURF is a tablet that is taken by mouth.
What are the benefits of this drug?
Patients treated with LONSURF lived an average of 7.1 months compared to 5.3 months for those who received a placebo pill.
On average, the time to disease progression (the amount of time from the start of treatment to when the cancer was found to be growing) was two months for patients on LONSURF compared to 1.7 months for patients receiving placebo.
What are the benefits of this drug (results of trials used to assess efficacy)?
The table and figure below summarize the primary efficacy outcome measure, overall survival (OS), and an additional efficacy outcome measure, progression-free survival (PFS), in the clinical trial.
| LONSURF | Placebo | |
|---|---|---|
| (N=534) | (N=266) | |
| Overall Survival | ||
| Number of deaths, N (%) | 364 (68) | 210 (79) |
| Median OS (months)a [95% CI]b | 7.1 [6.5, 7.8] | 5.3 [4.6, 6.0] |
| Hazard ratio [95% CI] | 0.68 [0.58, 0.81] | |
| P-valuec | ||
| Progression-Free Survival | ||
| Number of Progression or Death, N (%) | 472 (88) | 251 (94) |
| Hazard ratio [95% CI] | 0.47 (0.40, 0.55) | |
| P-valuec | ||
a Kaplan-Meier estimates
b Methodology of Brookmeyer and Crowley
c Stratified log-rank test (strata: KRAS status, time since diagnosis of first metastasis, region)
LONSURF Prescribing Information, Table 3
Figure 4. Kaplan-Meier Curves of Overall Survival
LONSURF Prescribing Information, Figure 1
Were there any differences in how well the drug worked in clinical trials among sex, race and age?
Subgroup analyses were conducted for sex, race and age.
- Sex: LONSURF was similarly effective in men and women.
- Race: LONSURF was similarly effective in whites and non-whites.
- Age: LONSURF was similarly effective in patients below the age of 65 years and those 65 years and above.
Were there any differences in how well the drug worked in clinical trials among sex, race and age groups?
The table below summarizes the efficacy subgroup analysis.
Table 3. Subgroup Analyses of Overall Survival: Gender, Age, and Race
|
Subgroups |
Events/N | Median PFS | HR (95% CI) | ||
|---|---|---|---|---|---|
| LONSURF | Placebo | LONSURF | Placebo | ||
|
Male |
220/326 | 128/165 | 7.3 | 5 | 0.69 (0.55, 0.87) |
|
Female |
144/208 | 82/101 | 6.8 | 5.6 | 0.68 (0.51, 0.90) |
|
Age |
203/300 | 113/148 | 7.1 | 5.7 | 0.74 (0.59, 0.94) |
|
Age ≥ 65 |
161/234 | 97/118 | 7 | 4.6 | 0.62 (0.48, 0.80) |
|
Caucasian |
185/306 | 115/155 | 6.3 | 4.9 | 0.66 (0.52, 0.83) |
|
Non-Caucasian |
179/228 | 95/111 | 7.8 | 5.8 | 0.71 (0.55, 0.92) |
PFS=Progression-Free Survival; HR=hazard ratio
Adapted from FDA Clinical Review
What are the possible side effects?
The most common side effects of treatment with LONSURF are low red blood cell count (anemia), a decrease in infection-fighting white blood cells (neutropenia) or blood platelets (thrombocytopenia), physical weakness, extreme tiredness and lack of energy (fatigue), nausea, decreased appetite, diarrhea, vomiting, and abdominal pain.
What are the possible side effects (results of trials used to assess safety)?
The table below summarizes adverse events in the clinical trial.
Table 4. Per Patient Incidence of Adverse Drug Reactions (≥5%) in the Trial Occurring More Commonly (>2%) than in Patients Receiving Placebo
Adverse Reactions |
LONSURF N=533 |
Placebo N=265 |
||
|---|---|---|---|---|
| All Grades | Grades 3-4* | All Grades | Grades 3-4* | |
| Gastrointestinal disorders | ||||
| Nausea | 48% | 2% | 24% | 1% |
| Diarrhea | 32% | 3% | 12% | |
| Vomiting | 28% | 2% | 14% | |
| Abdominal pain | 21% | 2% | 18% | 4% |
| Stomatitis | 8% | 6% | 0% | |
| General disorders and administration site conditions | ||||
| Asthenia/fatigue | 52% | 7% | 35% | 9% |
| Pyrexia | 19% | 1% | 14% | |
| Metabolism and nutrition disorders | ||||
| Decreased appetite | 39% | 4% | 29% | 5% |
| Nervous system disorders | ||||
| Dysgeusia | 7% | 0% | 2% | 0% |
| Skin and subcutaneous tissue disorders | ||||
| Alopecia | 7% | 0% | 1% | 0% |
*No Grade 4 definition for nausea, abdominal pain, or fatigue in National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE), version 4.03
LONSURF Prescribing Information, Table 1
Were there any differences in side effects among sex, race and age?
Subgroup analyses were conducted for sex, race and age.
- Sex: The risk of overall side effects was similar in men and women.
- Race: The risk of overall side effects was similar among whites and non-whites.
- Age: The risk of certain side effects of LONSURF was higher in patients 65 years of age or older compared to patients younger than 65 years. These side effects, related to specific levels of decreased counts of blood cells, are described in MORE INFO.
Were there any differences in side effects of the clinical trials among sex, race and age groups?
According to LONSURF Prescribing Information, patients 65 years of age or older who received LONSURF had a higher incidence of the following compared to patients younger than 65 years of age: Grade 3 or 4 neutropenia (48% vs 30%), Grade 3 anemia (26% vs 12%), and Grade 3 or 4 thrombocytopenia (9% vs 2%).
The following table summarizes adverse events by gender in the clinical trial.
Table 5. Summary of Adverse Events by Gender
| LONSURF N=533 n (%) |
Placebo N=265 n (%) |
|||
|---|---|---|---|---|
| Male N=326 |
Female N=207 |
Male N=164 |
Female N=101 |
|
| Subjects who experienced an AE | 322 (99) | 202 (98) | 152 (93) | 95 (94) |
| Subjects who experienced an SAE | 88 (27) | 68 (33) | 60 (37) | 28 (28) |
Adapted from FDA Clinical Review
The following table summarizes treatment-emergent adverse events by race.
Table 6. Analysis of Treatment Emergent Adverse Events by Race Group
| Race Group | LONSURF N=533 n (%) |
Placebo N=265 n (%) |
|---|---|---|
| White | 298 (97.7) | 144 (93.5) |
| Black or African American | 4 (100) | 5 (100) |
| Asian | 183 (99.5) | 87 (92.6) |
Analysis provided by company
WHO WAS IN THE CLINICAL TRIALS?
Who participated in the clinical trials?
The FDA approved LONSURF based on evidence from a clinical trial of 800 patients with metastatic colorectal cancer. The trial was conducted at 101 sites in 13 countries, including U.S., Sweden, Japan, Italy, Ireland, Great Britain, France, Spain, Germany, Czech Republic, Belgium, Austria, and Australia.
Figure 1 summarizes how many men and women were enrolled in the clinical trial.
Figure 1. Baseline Demographics by Sex
Clinical Trial Data
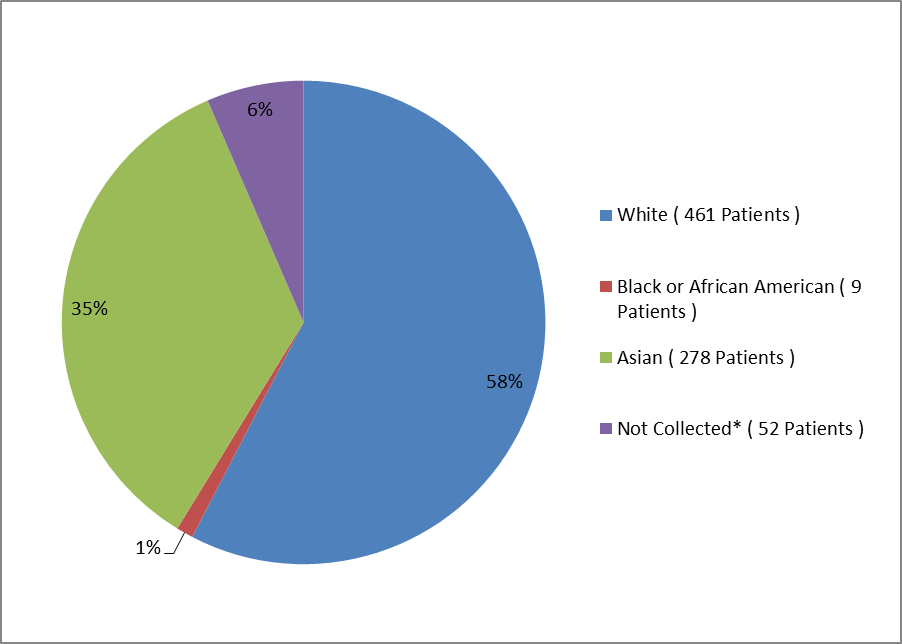
Figure 2 and Table 1 summarize how many patients by race were enrolled in the clinical trial.
Figure 2. Baseline Demographics by Race
*=collection of race data not permitted in all countries
Clinical Trial Data
Table 1. Demographics of Efficacy Trials by Race
| Race | Number of Patients | Percentage |
|---|---|---|
| White | 461 | 58% |
| Black or African American | 9 | 1% |
| Asian | 278 | 35% |
| Not Collected | 52 | 6% |
Clinical Trial Data
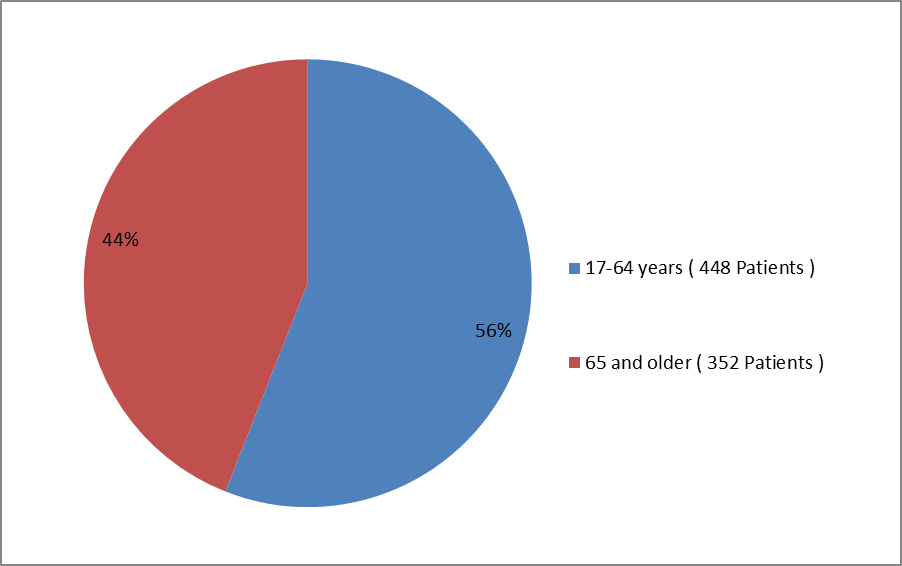
The figure below summarizes how many patients by age group were enrolled in the clinical trial.
Figure 3. Baseline Demographics by Age
Clinical Trial Data
Who participated in the trials?
The table below summarizes baseline demographics for the trial.
Table 7. Baseline Demographics
| Demographic | LONSURF | Placebo |
|---|---|---|
| N=534 | N=266 | |
| Age | ||
| Median age (range) | 63 (27-82) | 63 (27-82) |
| Mean age (±SD) | 62 (10) | 62 (11) |
| Age ≥ 65 years, n (%) | 234 (44) | 118 (44) |
| Age ≥ 75 years, n (%) | 36 (7) | 24 (9) |
| Gender | ||
| Male, n (%) | 326 (61) | 165 (62) |
| Female, n (%) | 208 (39) | 101 (38) |
| Race | ||
| White, n (%) | 306 (57) | 155 (58) |
| Black, n (%) | 4 (1) | 5 (2) |
| Asian, n (%) | 184 (34) | 94 (35) |
| Not collected, n (%) | 40 (7) | 12 (5) |
| Geographic Region | ||
| Region 1 (Japan), n (%) | 178 (33) | 88 (33) |
| Region 2 (U.S./Europe), n (%) | 356 (67) | 178 (67) |
FDA Clinical Review
How were the trials designed?
Patients were randomly assigned to receive either LONSURF plus best supportive care (BSC) or placebo plus BSC. The treatment continued until the disease progressed, the side effects became too toxic, or the patient decided to discontinue the study.
The clinical efficacy and safety of LONSURF were evaluated in an international, randomized, double-blind, placebo-controlled study conducted in patients with previously treated metastatic colorectal cancer (CRC).
Patients were randomized 2:1 to receive LONSURF plus best supportive care (BSC) or matching placebo plus BSC. Randomization was stratified by KRAS status (wild-type vs. mutant), time since diagnosis of first metastasis (18>
GLOSSARY
CLINICAL TRIAL: Voluntary research studies conducted in people and designed to answer specific questions about the safety or effectiveness of drugs, vaccines, other therapies, or new ways of using existing treatments.
COMPARATOR: A previously available treatment or placebo used in clinical trials that is compared to the actual drug being tested.
EFFICACY: How well the drug achieves the desired response when it is taken as described in a controlled clinical setting, such as during a clinical trial.
PLACEBO: An inactive substance or “sugar pill” that looks the same as, and is given the same way as, an active drug or treatment being tested. The effects of the active drug or treatment are compared to the effects of the placebo.
SUBGROUP: A subset of the population studied in a clinical trial. Demographic subsets include sex, race, and age groups.
PRESCRIBING INFORMATION