
Artificial Intelligence and Medical Products
Learn more about how the FDA is shaping the future of health care through the responsible and innovative integration of AI.

Learn more about how the FDA is shaping the future of health care through the responsible and innovative integration of AI.

You can help. Get the facts from the FDA. Stop the spread of false rumors, and share the facts with your loved ones.
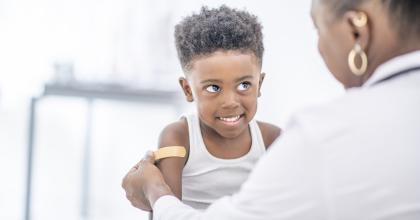
FDA-approved vaccines to prevent measles are safe and effective. Talk to your health care provider and learn more about how childhood vaccines can protect your children.
Alerting you to the latest recalls of potentially unsafe FDA-regulated products gathered from press releases and other public notices. Not all recalls have press releases or are posted on this website.
Apr 18
Food & Beverages
Apr 13
Animal & Veterinary
Apr 12
Food & Beverages

I recently had the privilege of official travel to the U.K. and European Union with our global affairs team.

At the FDA, we’ve been working for years to anticipate and prepare for the challenges of Artificial Intelligence (AI), and also to harness its potential.

FDA and the Office for Human Research Protections have published a draft guidance: “Key Information and Facilitating Understanding in Informed Consent.”
Apr 16
Drugs
Apr 03
Drugs
Apr 03
Dietary Supplements, Food & Beverages